A.4. Consider the system; NO4(g) 2NO2(g) Kp 11 at 100C. Starting with pure NO4 at a...
Fantastic news! We've Found the answer you've been seeking!
Question:
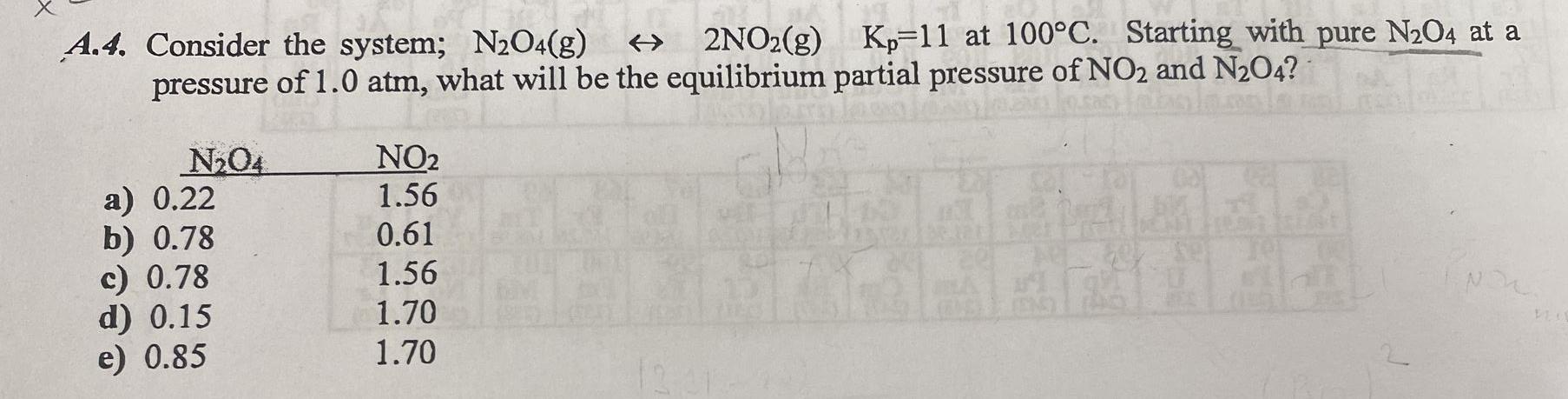
Transcribed Image Text:
A.4. Consider the system; N₂O4(g) 2NO2(g) Kp 11 at 100°C. Starting with pure N₂O4 at a pressure of 1.0 atm, what will be the equilibrium partial pressure of NO2 and N₂O4? N₂04 a) 0.22 b) 0.78 c) 0.78 d) 0.15 e) 0.85 NO₂ 1.56 0.61 1.56 1.70 1.70 Ca se e A.4. Consider the system; N₂O4(g) 2NO2(g) Kp 11 at 100°C. Starting with pure N₂O4 at a pressure of 1.0 atm, what will be the equilibrium partial pressure of NO2 and N₂O4? N₂04 a) 0.22 b) 0.78 c) 0.78 d) 0.15 e) 0.85 NO₂ 1.56 0.61 1.56 1.70 1.70 Ca se e
Expert Answer:
Answer rating: 100% (QA)
The detailed answer for the above question is provided below It seems like you are trying to solve a ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-6. On December 12, Irene purchased the building where her store is located. She paid...
-
A researcher wanted to find out if there was difference between older movie goers and younger movie goers with respect to their estimates of a successful actors income. The researcher first...
-
Expected manufacturing costs for Imperial Data Devices are as follows: Variable Costs Fixed Costs per Month Direct material $8.00/unit Supervisory salaries $17,000 Direct labor 3.50/unit Factory...
-
Prepare journal entries and post them to T-accounts for all transactions in Exercise 7-30, including requirement 2. Summarize how these journal entries differ from the normal-costing entries...
-
For each transition matrix P in Problems 2330, solve the equation SP = S to find the stationary matrix S and the limiting matrix P. P = .1 .9 .6 .4
-
The United States Census Bureau tracks shifts in population by each state and region in the United States. The net migration rate is an indicator of the movement of people from one area to another. A...
-
A hotel housekeeper, Alison Harvey, was observed five time son each of four task elements, as shown in the following table. On the basis of these observations, find the standard time for the process....
-
Discuss why the learning websites baby cant wait and naeyc are helpful and developmentally appropriate for the children with which you work?
-
Imax Corporation is a large entertainment technology company, with headquarters in New York and Toronto, and theatres worldwide. Its share price, which was as high as Can.$ 13.89 on the Toronto Stock...
-
Write down the Power-Interest Grid for Stakeholders.
-
Use the given information to find sin28, cos28, and tan28. 4. cose, and 0 < <90
-
Koyo, Inc. sold its 10% bonds with a maturity value of $7,000,000 on August 1, 20251 for $6,896,000, before paying $100,000 of bond issue costs. At the time of the sale the bonds had 5 years until...
-
Cathode ray tubes (CRTs) used in old-style televisions have been replaced by modern LCD and LED screens. Part of the CRT included a set of accelerating plates separated by a distance of about 1.38...
-
The Antarctic Circumpolar Current flows between the Palmer Peninsula (the northernmost part of the Antarctic) and the southern tip of South America at 2 m sec-1. If the flow is geostrophic, what is...
-
Discuss the design principles and implementation strategies behind fault tolerance mechanisms in operating systems, including redundancy, error detection, and error recovery techniques to ensure...
-
Selected hypothetical comparative statement data for the giant bookseller Barnes & Noble are presented here. All balance sheet data are as of the end of the fiscal year (in millions). 2022 2021 Net...
-
The area of square PQRS is 100 ft2, and A, B, C, and D are the midpoints of the sides. Find the area of square ABCD. B A
-
The second-order liquid-phase reaction C6H5COCH2Br + C6H5N C6H5COHCH2NC5H5Br is carried out in a batch reactor at 35C. The specific reaction-rate constant is 0.0445 dm 3 /mol/min. Reactor 1 is...
-
What if someone suggested you could use the solution to the flow-dispersion-reactor equation, Equation (18-18), for a second-order equation by linearizing the rate law by lettering rA=kCA2(kCA0/2)CA...
-
The elementary gas-phase reaction A B takes place isobarically and isothermally in a PFR where 63.2% conversion is achieved. The feed is pure A. It is proposed to put a CSTR of equal volume upstream...
-
When a contracting hot ball of gas spins into a disk shape, it cools faster due to (a) increased radiation transfer. (b) increased surface area. (c) decreased insulation. (d) increased convection...
-
The Sun contains what percentage of the solar systems mass? (a) about 35% (b) 85% (c) the percentage varies over time (d) over 99%
-
The nebular theory is based on the observation that the solar system (a) follows patterns indicating that it formed progressively from physical processes. (b) has a structure much like an atom. (c)...

Study smarter with the SolutionInn App


