Question: All and Especially problem number 4 pleas . Problem 1 Convert the following quantities as shown below Ix=2*10 mA Energy 11000 mAh EA 200 kWh

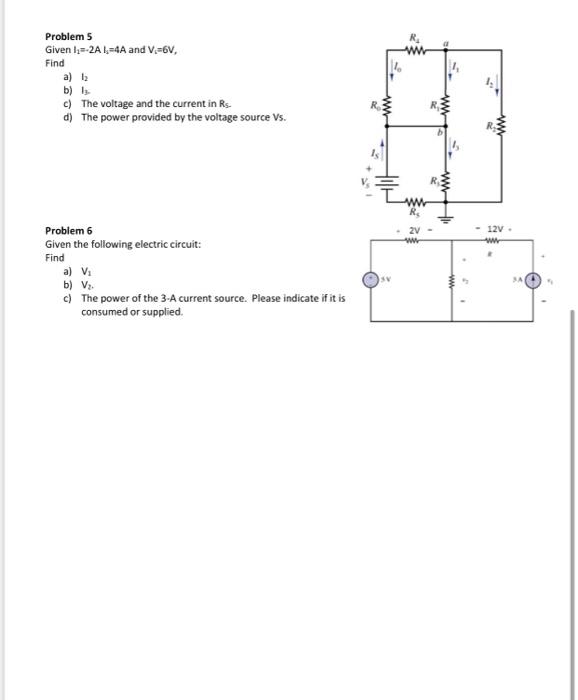
Problem 1 Convert the following quantities as shown below Ix=2*10 mA Energy 11000 mAh EA 200 kWh V2-0.0132MV Rc 1.70MQ Time 2 Hours 20 minutes PA-4400mW Energy 380 kJ | RA=2.7x10 Problem 3 A 12 V Battery has a nominal rate of 200 A-h. Find: W A Ah J kv Seconds Problem 2: Complete the following table: a) Given P = 15 W and V=6V, find the current I b) Given Energy-6000 Wh and P-120 W, find the time t c) Given V 220 V and P=300W, find the resistance R d) Given V 220 V and P=300W, find the current I e) Given V=110 V and R=20 $2, find the current I and the Power P f) Given V=220 V and I = 2A, find the power P W Wh 52 a) The charge potentially derived from the battery b) The number of electrons contained in this charge found in part (a) Problem 4 Given the following voltage and current rate change for a battery a) Find the charge transfer to the battery in 10 hours. b) Find the energy transferred to the battery in 10 hours. Hint: Use the following formula: Q=i(1)dt To w=p(t)dt = v(n)i(r)dr Battery voltage Battery current 10mA 0 5h 10% ION F Problem 5 Given I=-2A 1,-4A and V, 6V, Find a) b) Is c) The voltage and the current in Rs. d) The power provided by the voltage source Vs. Problem 6 Given the following electric circuit: Find a) V b) V. c) The power of the 3-A current source. Please indicate if it is consumed or supplied. SV 2V www - 12V. ww 3A
Step by Step Solution
3.46 Rating (149 Votes )
There are 3 Steps involved in it

Get step-by-step solutions from verified subject matter experts


