Problem #2 (20 points) Three quarts of water at 25 C is poured over 1.5 kilograms...
Fantastic news! We've Found the answer you've been seeking!
Question:
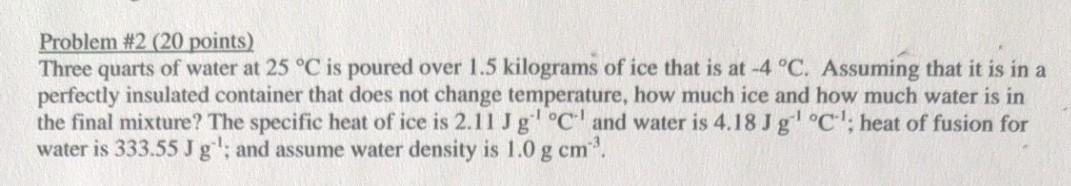
Transcribed Image Text:
Problem #2 (20 points) Three quarts of water at 25 °C is poured over 1.5 kilograms of ice that is at -4 °C. Assuming that it is in a perfectly insulated container that does not change temperature, how much ice and how much water is in the final mixture? The specific heat of ice is 2.11 J g¹ °C and water is 4.18 Jg¹ °C¹; heat of fusion for water is 333.55 J g'; and assume water density is 1.0 g cm³. Problem #2 (20 points) Three quarts of water at 25 °C is poured over 1.5 kilograms of ice that is at -4 °C. Assuming that it is in a perfectly insulated container that does not change temperature, how much ice and how much water is in the final mixture? The specific heat of ice is 2.11 J g¹ °C and water is 4.18 Jg¹ °C¹; heat of fusion for water is 333.55 J g'; and assume water density is 1.0 g cm³.
Expert Answer:
Related Book For 

Introduction to Chemical Engineering Thermodynamics
ISBN: 978-0071247085
7th edition
Authors: J. M. Smith, H. C. Van Ness, M. M. Abbott
Posted Date:
Students also viewed these physics questions
-
The unit of magnetic flux is named for Wilhelm Weber. The practical-size unit of magnetic field is named for Johann Karl Friedrich Gauss. Both were scientists at Göttingen, Germany....
-
The diffusion coefficient of glucose in water at 25 C is 6.73 10 10 m 2 s 1 . Estimate the time required for a glucose molecule to undergo a root-mean-square displacement of 5.0mm.
-
An ideal monatomic gas is held in a perfectly insulated cylinder fitted with a movable piston. The initial pressure of the gas is 110 kPa, and its initial temperature is 280 K. By pushing down on the...
-
Part 2: Use the activity-based information below to calculate the cost of each unit of product using machine setups instead of machine hours. Cost Driver Number of Machine Setups Operating Machines...
-
Table provides a vendor comparison for a system that is under consideration for purchase. Based on the data provided, which system should be selected under each of the following scenarios? a. Lowest...
-
Your roommate drops a tennis ball from a third-story balcony. It hits the sidewalk and bounces as high as the second story. Draw a motion diagram, using the particle model, showing the ball 's...
-
Use the same facts as problem 20 and prepare entries using the following independent assumptions: a. Lease A is abandoned in 2022. b. Lease A is proved in 2022. c. Insignificant Lease Y, with a cost...
-
Current Liabilities and Ratios Several accounts that appeared on Kruses 2010 balance sheet are as follows: Required 1. Prepare the Current Liabilities section of Kruses 2010 balance sheet. 2. Compute...
-
(a) Let3+ and 2 = a + bi be complex numbers. Suppose that 7 Argument = 12' find Argument(22). 7-2 (b) Let the map f: CC be defined by f(z) = Find f() if=1+2i. 1 (c) Solve the equation -12 i(9-2),...
-
Jim and Joel have come to you to get an appraisal to lease their property. Using the information below, complete the following questions: Answer the below questions according to the property details...
-
Sawchuk Company is considering an important capital project it will raise external capital to finance. The firm is financed in part by equity investors and in part by rolling over $500 million par...
-
Draw as many resonance structures as you can for (a) The form of fluorescein present at pH = 9, and (b) The fluorescent group of the green fluorescent protein. Use the curved-arrow notation to derive...
-
In 2017, Apple told its suppliers to prepare for the sale of 100 million iPhones; however, in 2018 Apple updated its projections and informed suppliers that it expected iPhone demand to be lower....
-
Using white and black circles to represent different kinds of atoms, make a drawing that accurately represents each sample of matter: A solid element, a liquid compound, and a heterogeneous mixture....
-
(a) Draw the structure of P 4 S 3 and describe an appropriate bonding scheme for this molecule. Compare the structures of P 4 S 10 , P 4 S 3 and P 4 , and comment on the formal oxidation states of...
-
Butcher Enterprises has experienced substantial employee turnover among its office workers. During exit interviews, more than 80 percent stated that low pay was the top reason for resigning. The...
-
For a construction project, the main activities are shown in table (3), the following is required: I. Using the precedence diagram method, draw the network for the activities given in the following...
-
Using thermodynamic data from Appendix 4, calculate G at 258C for the process: 2SO 2 (g) + O 2 (g) 88n 2SO 3 (g) where all gases are at 1.00 atm pressure. Also calculate DG8 at 258C for this same...
-
Air at 1(aim) and 35C is compressed in a staged reciprocating compressor (with inter cooling) to a final pressure of 50(atm). For each stage, the inlet gas temperature is 35oC and the maximum...
-
A stream of water at 85oC, flowing at the rate of 5 kg s-l is formed by mixing water at 24oC with saturated steam at 400 kPa. Assuming adiabatic operation, at what rates are the steam and water fed...
-
A steam turbine operates adiabatically at a power level of 3,500 kW, Steam enters the turbine at 2,400 kPa and 500oC and exhausts from the turbine as saturated vapor at 20 kPa. What is the steam rate...
-
Last year, Miley decided to terminate the S corporation election of her solely owned corporation on October 17, 2018 (effective immediately), in preparation for taking it public. At the time of the...
-
You have been assigned to compute the income tax provision for Motown Memories Inc. (MM) as of December 31, 2019. The companys income statement for 2019 is provided below: MM prepared the following...
-
Jane has been operating Mansfield Park as a C corporation and decides she would like to make an S election. What is the earliest the election will become effective under each of these alternative...

Study smarter with the SolutionInn App



