An aqueous solution of 0.216 M ethanol. CH3CHOH, has a density of 0.996 g/mL. The percent...
Fantastic news! We've Found the answer you've been seeking!
Question:
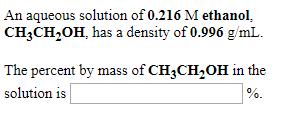
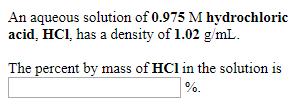

Transcribed Image Text:
An aqueous solution of 0.216 M ethanol. CH3CH₂OH, has a density of 0.996 g/mL. The percent by mass of CH3CH₂OH in the solution is %. An aqueous solution of 0.975 M hydrochloric acid, HCI, has a density of 1.02 g/mL. The percent by mass of HCI in the solution is %. An aqueous solution is 22.0% by mass ethanol, CH3CH₂OH, and has a density of 0.966 g/mL. The molarity of ethanol in the solution is M. An aqueous solution of 0.216 M ethanol. CH3CH₂OH, has a density of 0.996 g/mL. The percent by mass of CH3CH₂OH in the solution is %. An aqueous solution of 0.975 M hydrochloric acid, HCI, has a density of 1.02 g/mL. The percent by mass of HCI in the solution is %. An aqueous solution is 22.0% by mass ethanol, CH3CH₂OH, and has a density of 0.966 g/mL. The molarity of ethanol in the solution is M.
Expert Answer:
Answer rating: 100% (QA)
mass 1 Melavity molar mass molarity 2 1000 V in ml oxus x den... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
The percent by mass of bicarbonate (HCO3-) in a certain Alka-Seltzer product is 32.5 percent. Calculate the volume of CO2 generated (in mL) at 37C and 1.00 atm when a person ingests a 3.29-g tablet....
-
An aqueous solution of ammonium nitrite, NH4NO2, decomposes when heated to give off nitrogen, N2. NH4NO2(s) 2H2O(g) + N2(g) This reaction may be used to prepare pure nitrogen. How many grams of...
-
An aqueous solution of a molecular compound freezes at -0.086C. What is the molality of the solution?
-
Jaclyn Hargrove is the owner of six Pickwick Restaurants. For the past 10 years, she has always relied on her accountant to analyze her financial statements. Jaclyn feels that if she were able to...
-
What does the following statement mean? Indeed, every decision we make involves some kind of trade-off. Under what condition would the statement not be true? Does the entrepreneur face a tradeoff...
-
In Problems 107118, find the value of each expression if x = 2 and y = 1. 2 (x)
-
Describe some examples of autonomous robots.
-
Cost data for Bedford Manufacturing company for the month ending May 31, 2012, as follows: a. Prepare a cost of goods manufactured statement for May 2012. b. Determine the cost of goods sold for...
-
Calculate the activity in millicuries of 123I produced by the 121Sb (, 2n) 123I reaction, when 200-mg natural antimony (natural abundance of 121Sb is 57.3%) is irradiated for 2 h with an -particle...
-
Apperson and Fitz is a chain of clothing stores that caters to high school and college students. It publishes a quarterly catalogue and operates a Web site that features provocatively attired males...
-
2. A biologist examines the organelles present in the root cells, stem cells, and leaf cells of a green plant. She finds a high density of mitochondria and no chloroplasts in the root cells, but an...
-
last year Ivanhoe sold 42000 ankle support. the marketing manger is convinced that a 10% reduction in the sales price, combined with a $51000 increase in advertising. Will result in a 330% increase...
-
If alternative financial performance measures have been disclosed, has their relevance been satisfactorily explained? List non-GAAP measures used in the MD&A.
-
Beat, Inc., expects to sell 60,000 athletic uniforms for $80 each in 2012. Direct materials costs are $20, direct manufacturing labor is $8, and manufacturing overhead is $6 for each uniform. The...
-
Choose a video (youtube or TedTalk) that interests you and relates to communication and business. https://youtu.be/Z3HJCQJ2Lmo Summarize the talk Third, in one good paragraph explain why this talk...
-
What is the definition of combination pay and how does combination pay work for an employee with a Base Salary and Commission? What would be an example of an employee who has a Salary plus Commission...
-
Create a short program that demonstrates one of the functions shown below: Date class: long getTime() Random class: set the seed and generate a random number Date class: toString() Date class:...
-
Find the center of mass of a thin triangular plate bounded by the y-axis and the lines y = x and y = 2 - x if (x, y) = 6x + 3y + 3.
-
Confidence interval for calibration curve. To use a calibration curve based on n points, we measure a new value of y and calculate the corresponding value of x. The one-standard deviation uncertainty...
-
The oxidation numbers of Cu and Bi in high-temperature superconductors of the type Bi 2 Sr 2 (Ca 0.8 Y 0. 2)Cu 2 O x (which could contain Cu 2+ , Cu 3+ , Bi 3+ , and Bi 5+ ) can be measured by the...
-
What is the purpose of the trap in Figure 2-17 and the watchglass in Figure 2-20? Figure 2-17 Figure 2-20 Gooch filter crucible To air -Rubber adaptor -Glass funnel To house vacuum Porous glass Gooch...
-
Cooling pipes at three nuclear power plants are investigated for deposits that would inhibit the flow of water. From 30 randomly selected spots at each plant, 13 from the first plant, 8 from the...
-
With reference to Exercise 10.57, find a large sample 95% confidence interval for the true difference of the probabilities of failure. Data From Exercise 10.57 10.57 Two bonding agents, A and B, are...
-
Two hundred tires of each of four brands are individually placed in a testing apparatus and run until failure. The results are obtained the results shown in the following table: (a) Use the 0.01...

Study smarter with the SolutionInn App


