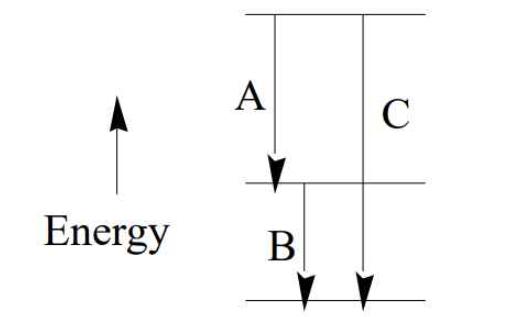
An atom of some element X has three electronic transitions, A, B, and C. Suppose that it
Fantastic news! We've Found the answer you've been seeking!
Question:
An atom of some element X has three electronic transitions, A, B, and C. Suppose that it has been found that the wavelength for transition A is 459.1 nm and that for B is 577.9 nm. (a) What is the energy (in J) for a photon given off in transition C? (b) What is the wavelength (in nm) for transition C?
Related Book For 

Posted Date:




