An ideal gas has pressure po, volume V, and temperature To. It is taken through an...
Fantastic news! We've Found the answer you've been seeking!
Question:
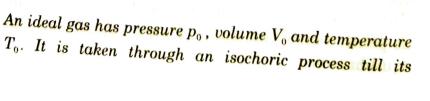

Transcribed Image Text:
An ideal gas has pressure po, volume V, and temperature To. It is taken through an isochoric process till its pressure is doubled. It is now isothermally expanded to get the original pressure. Finally, the gas is isobarically compressed to its original volume V. (a) Show the process on a p-V diagram. (b) What is the temperature in the isothermal part of the process? (c) What is the volume at the end of the isothermal part of the process? An ideal gas has pressure po, volume V, and temperature To. It is taken through an isochoric process till its pressure is doubled. It is now isothermally expanded to get the original pressure. Finally, the gas is isobarically compressed to its original volume V. (a) Show the process on a p-V diagram. (b) What is the temperature in the isothermal part of the process? (c) What is the volume at the end of the isothermal part of the process?
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these physics questions
-
An ideal gas has an adiabatic exponent y. In some process its molar heat capacity varies as C = a/T, where a is a constant. Find: (a) The work performed by one mole of the gas during its heating from...
-
An ideal gas has a molar heat capacity Cv at constant volume. Find the molar heat capacity of this gas as a function of its volume V, if the gas undergoes the following process: (a) T = Toe v ; (b) p...
-
An ideal gas has a gas constant R = 0.3 kJ/kgK and a constant-volume specific heat cv = 0.7 kJ/kgK. If the gas has a temperature change of 100C, choose the correct answer for each of the following:...
-
Explain what quality management knowledge and skills an auditor should possess when auditing
-
What is the reason behind the use of the minimum ratio test in selecting the pivot row? What might happen without it?
-
Brainstorm as many examples of social media as you can and write these down in a list. Use Cavazza's (2012) Social Media Landscape model to categorize each of the examples you have listed. Which of...
-
Generalized Regression Techniques and Variable Selection In Chapter 9, we introduced generalized regression techniques as an approach to handling the multicollinearity problem. The LASSO can...
-
BrasPetro S.A., a large Brazilian petrochemical company, reported a balance of R$1,594.9 million in Accounts Receivable at the beginning of Year 7 and R$1,497.0 million at the end of Year 7. Its...
-
Tegan is a commission sales person. In October, Tegan sold $50,000 in product. What is the value of Tegan's commission? The commission rates are: 2% for the first $10,000 4% for the next $15,000 ...
-
Use the same scale to construct boxplots for the full IQ scores (IQF) for the low lead level group (group 1) and the high lead level group (group 3) in Data Set 7 "IQ and Lead" in Appendix B. Use the...
-
Jo and Kim are best friends, they both want to start investing more in order to retire in 8 years. They both have a risk profile of moderate to balanced investor, as they are in there early 50s but...
-
A person with an SAT score of 2200 is twice as qualified for college as a person with a score of 1100. complete classification. Determine whether the given statement represents a meaningful ratio, so...
-
Write a brief description of some ways in which you think that the science of statistics can be used in your major field of study. (If you have not yet selected a major, answer the question for a...
-
A clinical trial involved the use of a nicotine gum as an aid to help smokers stop smoking. The clinical trial involved 1,000 college students who were paid for their participation, and the results...
-
Wind with a speed of 40 mi/h moves four times as fast as wind with a speed of 10 mi/h. Determine whether the given statement represents a meaningful ratio, so that the ratio level of measurement...
-
What is the margin of error in a statistical study, and why is it important? Does It make Sense?
-
On average, 60% of Caf Vert's sales are for immediate cash settlement, with the remaining 40% being for credit. Of these credit sales, 50% are normally received in the month of sale, 25% in the...
-
Halley's comet travels in an ellipti- cal orbit with a = 17.95 and b = 4.44 and passes by Earth roughly every 76 years. Note that each unit represents one astronomical unit, or 93 million miles. The...
-
A monochromatic beam of X-rays with a wavelength of 0.280 nm is scattered by a metal foil. If the scattered beam has a wavelength of 0.281 nm, what is the observed scattering angle?
-
In a simulated head-on crash test, a car impacts a wall at 25 mi/h (40 km/h) and comes abruptly to rest. A 120-lb passenger dummy (with a mass of 55 kg), without a seatbelt, is stopped by an air bag,...
-
A large square (finite size) flat plate is uniformly positively charged and in the horizontal plane. Determine the direction of the electric field at the following locations: (a) Just above the...
-
Enter up a columnar purchases day book with columns for the various expenses for J. Still for the month from the following information on credit items. 2016 January f 1 Bought goods from H. Graham...
-
A Enter up a columnar purchases day book with columns for the various expenses for F. Graham for the month from the following information on credit items. 2016 June Bought goods from J. Syme 4 Bought...
-
Enter up the relevant accounts in the purchases and general ledgers from the columnar purchases day book you completed for Review Question 20.4A. Data From Review Question 20.4A 20.4A Enter up a...

Study smarter with the SolutionInn App



