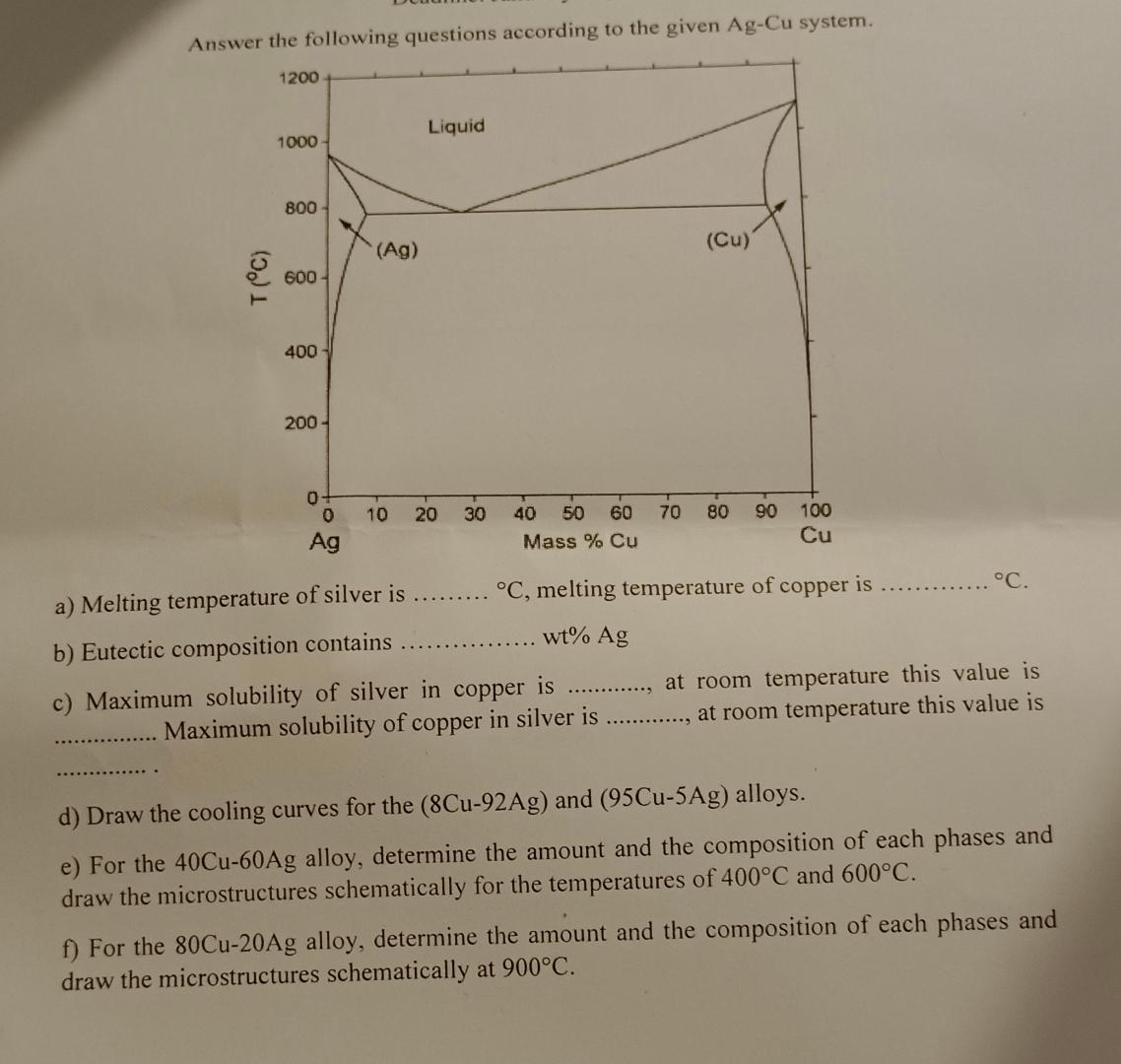
Answer the following questions according to the given Ag-Cu system. 1200 T (C) 1000- 800 600-...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Answer the following questions according to the given Ag-Cu system. 1200 T (C) 1000- 800 600- 400 200- 0 Ag (Ag) 10 Liquid 20 30 40 50 60 Mass % Cu 70 (Cu) 80 90 100 Cu C, melting temperature of copper is wt% Ag C. a) Melting temperature of silver is b) Eutectic composition contains is copper at room temperature this value is c) Maximum solubility of silver in Maximum solubility of copper in silver is ............., at room temperature this value is d) Draw the cooling curves for the (8Cu-92Ag) and (95Cu-5Ag) alloys. e) For the 40Cu-60Ag alloy, determine the amount and the composition of each phases and draw the microstructures schematically for the temperatures of 400C and 600C. f) For the 80Cu-20Ag alloy, determine the amount and the composition of each phases and draw the microstructures schematically at 900C. Answer the following questions according to the given Ag-Cu system. 1200 T (C) 1000- 800 600- 400 200- 0 Ag (Ag) 10 Liquid 20 30 40 50 60 Mass % Cu 70 (Cu) 80 90 100 Cu C, melting temperature of copper is wt% Ag C. a) Melting temperature of silver is b) Eutectic composition contains is copper at room temperature this value is c) Maximum solubility of silver in Maximum solubility of copper in silver is ............., at room temperature this value is d) Draw the cooling curves for the (8Cu-92Ag) and (95Cu-5Ag) alloys. e) For the 40Cu-60Ag alloy, determine the amount and the composition of each phases and draw the microstructures schematically for the temperatures of 400C and 600C. f) For the 80Cu-20Ag alloy, determine the amount and the composition of each phases and draw the microstructures schematically at 900C.
Expert Answer:
Answer rating: 100% (QA)
Solutions Step 1 a Melting point temperature of silvery Tag temperature 9619C of copperTag1085 6 Eut... View the full answer

Related Book For 

Posted Date:
Students also viewed these mechanical engineering questions
-
a. Calculate the project's initial Year O cash flow, taking into account all side effects. Assume that the net working capital will not require flotation costs. (A negative answer should be indicated...
-
Greenwood Ltd., one of the largest commercial builders in the city, has decided to purchase a new piece of equipment due to a very strong order book.You have just been hired as their new financial...
-
The copper-silver phase diagram is shown in Figure 11-30. Copper has a higher melting point than silver. Refer to the silver-rich solid phase as gamma (γ) and the copper-rich solid phase...
-
You have just completed your four-year degree at LLC University! Your student loans that you have accumulated while studying at LLC total $25,000. Since you have graduated, you must now begin...
-
Find the condition number of the following matrices. Which would you characterize as ill conditioned? (a)_ 3 999 .341 1.001 .388 o(L01 29997) (d) (-1 2-4) )1.00 1.9997 2 10 6 72 96 103 2 (e) 4255 59...
-
A portfolio is made up of two assets, A and B, whose volatilities are equal to, respectively, 55% and 35%. The stock weightings in the portfolio are as follows: 40% in A and 60% in B. What is the...
-
For accounting purposes, how are liabilities defined?
-
Ace Hardware is a retailer-owned cooperative, with 4,600 hardware, home center, and building materials stores. At the time of this case, Ace was a private company that was planning to go public. In...
-
15.12 A Northern British Columbia lumberyard has four jobs on order, as shown in the following table. Today is day 205 on the yard's schedule. Job Due Date Remaining Time (days) A 212 6 B 209 3 C 208...
-
Which of the following are correct? (A) If = 0 for real gas need not be 0. then OP (B) For a reversible reaction at equilibrium, the entropy of the system is maximum while that of the surrounding is...
-
What are some political arguments regarding slavery in the following documents ? - Frederick Douglass Dred Scott v. Sanford (1857) -Charles Summer The Crime of Kansas (1856) -Southern Press Review...
-
How did mobilization for World War II impact Texas and contribute towards modernization? How did Texas change as a result of the Civil Rights Movement? Describe 3-4 major aspects of modern Texas.
-
A manufacturer's contribution margin income statement for the year follows. Prepare a contribution margin income statement if the number of units sold (a) increases by 200 units and (b) decreases by...
-
. A student used a different calibration curve to determine the mass of sugar in a 100.0 mL sample of Gatorade to be 6.95 g. How many grams sugar would be in a 12 oz. bottle of this Gatorade?
-
Consultants notify management of Discount Pharmaceuticals that a stroke medication poses a potential health hazard. Counsel indicates a product recall is probable and is estimated to cost the company...
-
How can TV series and films in South Africa drive tourism locally and globally?
-
Let (x) = x 2 - 9, g(x) = 2x, and h(x) = x - 3. Find each of the following. (((--) 2
-
Give equations for the formation and hydrogenolysis of glutamine benzyl ester.
-
(a) Nitrogen has relatively stable isotopes (half-life greater than 1 second) of mass numbers 13, 14, 15, 16, and 17. (All except 14N and 15N are radioactive.) Calculate how many protons and neutrons...
-
Many of the condensations we have studied are reversible. The reverse reactions are often given the prefix retro-, the Latin word meaning backward. Propose mechanisms to account for the following...
-
Decision scope categorizes decisions into three broad areas of managerial decision making. These three areas are operational control, management control, and strategic planning. In your own words,...
-
Write a two-page report explaining the two basic strategies that S&S, Inc. can pursue and the three different strategic positions that it can adopt. Include in your report a discussion and examples...
-
Information technology is continually changing the nature of accounting and the role of accountants. Write a two-page report describing what you think the nature of the accounting function in a large...

Study smarter with the SolutionInn App


