Arrange the following molecules according to the strength of their dispersion forces. Dispersion Forces Molecules (6...
Fantastic news! We've Found the answer you've been seeking!
Question:
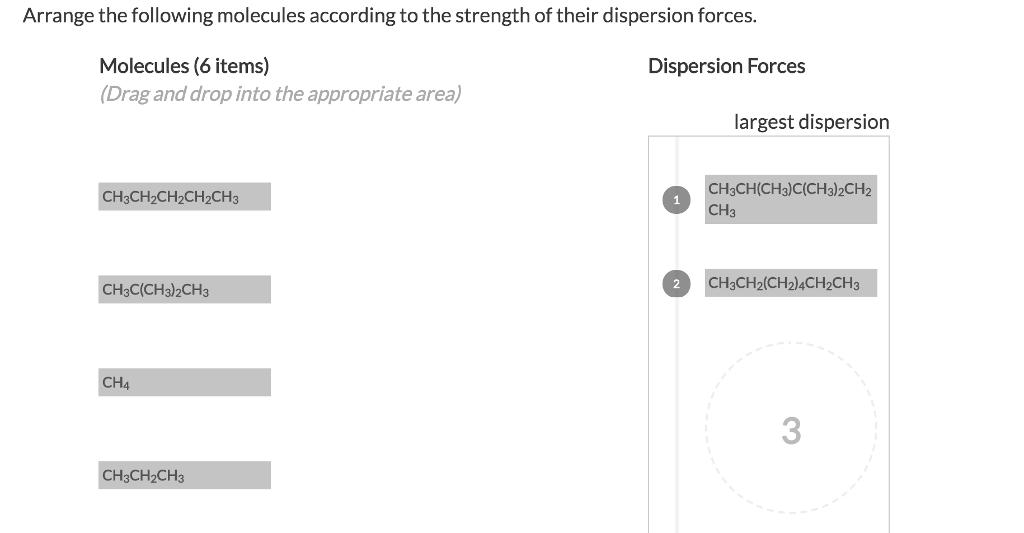
Transcribed Image Text:
Arrange the following molecules according to the strength of their dispersion forces. Dispersion Forces Molecules (6 items) (Drag and drop into the appropriate area) CH3CH₂CH₂CH₂CH3 ||| CH3C(CH3)2CH3 CH4 CH3CH₂CH3 2 largest dispersion CH3CH(CH3)C(CH3)2CH2 CH3 CH3CH2(CH2)4CH₂CH3 3 Arrange the following molecules according to the strength of their dispersion forces. Dispersion Forces Molecules (6 items) (Drag and drop into the appropriate area) CH3CH₂CH₂CH₂CH3 ||| CH3C(CH3)2CH3 CH4 CH3CH₂CH3 2 largest dispersion CH3CH(CH3)C(CH3)2CH2 CH3 CH3CH2(CH2)4CH₂CH3 3
Expert Answer:
Answer rating: 100% (QA)
London dispersion forces These are interactive forces that exists between the atoms of nonpola... View the full answer

Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Posted Date:
Students also viewed these accounting questions
-
6) What is the equation of the parent function of g(x) = -4? a) g(x) = 2* b) g(x) = x c) g(x) = - (x)6 (p
-
Bond Strength Homework Unanswered Arrange the following molecules from least stable (highest or least negative potential energy at the top) to most stable (lowest or most negative potential energy at...
-
Name the following molecules according to the IUPAC nomenclature system. (a) (b) (c) (d) (e) (f) CH32 Cl CH3 Br
-
In an organization, leaders treat employees as ends when they ______. Multiple choice question. restrict employees' choices allow employees to create their own purposes assume that the employees'...
-
Suppose a recent income statement for McDonalds Corporation shows cost of goods sold $4,852.7 million and operating expenses (including depreciation expense of $1,201 million) $10,671.5 million. The...
-
Basic capital budgeting problem with straight-line depreciation. The Roberts Company has cash inflows of $140,000 per year on project A and cash outflows of $100,000 per year. The investment outlay...
-
Which inventory costing method assigns the newest, most recent, costs incurred during the period to ending inventory? a. Specific-unit cost b. First-in, first-out (FIFO) C. Last-in, first-out (LIFO)...
-
Seattle Roast Coffee Company produces Columbian coffee in batches of 8,000 pounds. The standard quantity of materials required in the process is 8,000 pounds, which cost $5.00 per pound. Columbian...
-
1. How does Supply-Chain Tech help Direct-to-Consumer Sales companies gain competitive advantages? 2. For companies like PepsiCo Inc. and Nike, who transition to e-commerce- what type of technologies...
-
The Sally Coffee Shop is considering closing one hour earlier in the evening through the week. Sally obtained the following data from a study taken last week of the number of customers between the...
-
Slide #21: how much is v? Quoted price versus invoice price Today is 10/30/2030. A Treasury bond has a face value of $1000, maturity at 8/15/2032, coupon rate of 6%, and yield to maturity (BEY) 6.20%...
-
Alatorre Corporation, which manufactures shoes, hired a recent college graduate to work in its accounting department. On the first day of work, the accountant was assigned to total a batch of...
-
Assume the same information as in E14-4, except that the bonds were issued at 84.95 to yield 12%. Instructions Prepare the journal entries to record the following. (Round to the nearest euro.) (a)...
-
Allegro Supply Company, a newly formed corporation, incurred the following expenditures related to Land, to Buildings, and to Equipment. Instructions Determine the amounts that should be debited to...
-
Shlee Corporation issued a 4-year, 60,000, zero-interest-bearing note to Garcia Company on January 1, 2015, and received cash of 60,000. In addition, Shlee agreed to sell merchandise to Garcia at an...
-
On January 1, 2015, McLean Company makes the two following acquisitions. 1. Purchases land having a fair value of 300,000 by issuing a 5-year, zero-interest-bearing promissory note in the face amount...
-
How does an individual's identification with their role influence their level of engagement and commitment?
-
-x/2 x/4 If A = -x/2 and A-1 =6 then x equals
-
Using the information in Table 11-4 match up each set of the following IR signals with one of these naturally occurring compounds, camphor, menthol, chrysanthemic ester, epiandrosterone you can find...
-
Write the structures of the aldol condensation products of (a) pentanal; (b) 3-methylbutanal; (c) cyclopentanone.
-
Which of the following compounds are chiral? (a) 2-Methylheptane (b) 3-Methylheptane (c) 4-Methylheptane (d) 1,1-Dibromopropane (e) 1,2-Dibromopropane (f) 1,3-Dibromopropane (g) Ethene, H2C=CH2 (h)...
-
A car is modeled as shown in Fig. 6.23. Derive the equations of motion using Newton's second law of motion. Figure 6.23:- x(t) F(t) 21 31 Rigid bar, mass- 2m A G X3(1) F3(t) 5m x2(1) F(t) FIGURE 6.20...
-
Derive the equations of motion, using Newton's second law of motion, for each of the systems shown in Figs. 6.19. 8(t) 2k 2m k 4 M,(t) 000 3k Free Rigid bar, mass = 2m T x(1) F(t) m I X2(1) t F(1)...
-
The equations of motion derived using the displacements of the masses, \(x_{1}, x_{2}\), and \(x_{3}\) as degrees of freedom in Fig. 6.12 (Example 6.10) lead to symmetric mass and stiffness matrices...

Study smarter with the SolutionInn App


