Assume the market portfolio consists only of three stocks as follows: Stock Emperor King Sultan Current...
Fantastic news! We've Found the answer you've been seeking!
Question:
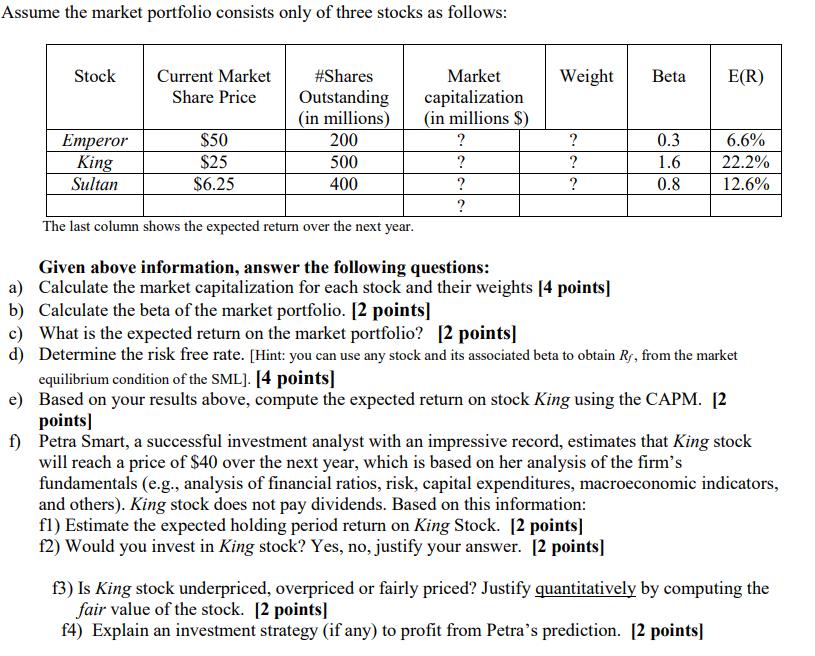
Transcribed Image Text:
Assume the market portfolio consists only of three stocks as follows: Stock Emperor King Sultan Current Market Share Price $50 $25 $6.25 # Shares Outstanding (in millions) 200 500 400 Market capitalization (in millions $) ? ? ? ? Weight Beta ? ? ? The last column shows the expected return over the next year. Given above information, answer the following questions: a) Calculate the market capitalization for each stock and their weights [4 points] b) Calculate the beta of the market portfolio. [2 points] 0.3 1.6 0.8 E(R) 6.6% 22.2% 12.6% c) What is the expected return on the market portfolio? [2 points] d) Determine the risk free rate. [Hint: you can use any stock and its associated beta to obtain Ry, from the market equilibrium condition of the SML]. [4 points] e) Based on your results above, compute the expected return on stock King using the CAPM. [2 points] f) Petra Smart, a successful investment analyst with an impressive record, estimates that King stock will reach a price of $40 over the next year, which is based on her analysis of the firm's fundamentals (e.g., analysis of financial ratios, risk, capital expenditures, macroeconomic indicators, and others). King stock does not pay dividends. Based on this information: fl) Estimate the expected holding period return on King Stock. [2 points] f2) Would you invest in King stock? Yes, no, justify your answer. [2 points] f3) Is King stock underpriced, overpriced or fairly priced? Justify quantitatively by computing the fair value of the stock. [2 points] f4) Explain an investment strategy (if any) to profit from Petra's prediction. [2 points] Assume the market portfolio consists only of three stocks as follows: Stock Emperor King Sultan Current Market Share Price $50 $25 $6.25 # Shares Outstanding (in millions) 200 500 400 Market capitalization (in millions $) ? ? ? ? Weight Beta ? ? ? The last column shows the expected return over the next year. Given above information, answer the following questions: a) Calculate the market capitalization for each stock and their weights [4 points] b) Calculate the beta of the market portfolio. [2 points] 0.3 1.6 0.8 E(R) 6.6% 22.2% 12.6% c) What is the expected return on the market portfolio? [2 points] d) Determine the risk free rate. [Hint: you can use any stock and its associated beta to obtain Ry, from the market equilibrium condition of the SML]. [4 points] e) Based on your results above, compute the expected return on stock King using the CAPM. [2 points] f) Petra Smart, a successful investment analyst with an impressive record, estimates that King stock will reach a price of $40 over the next year, which is based on her analysis of the firm's fundamentals (e.g., analysis of financial ratios, risk, capital expenditures, macroeconomic indicators, and others). King stock does not pay dividends. Based on this information: fl) Estimate the expected holding period return on King Stock. [2 points] f2) Would you invest in King stock? Yes, no, justify your answer. [2 points] f3) Is King stock underpriced, overpriced or fairly priced? Justify quantitatively by computing the fair value of the stock. [2 points] f4) Explain an investment strategy (if any) to profit from Petra's prediction. [2 points]
Expert Answer:
Answer rating: 100% (QA)
a To calculate the market capitalization and weights for each stock 1 Emperor Market capitalization Current Market Share Price Shares Outstanding Mark... View the full answer

Related Book For 

Intermediate Accounting
ISBN: 978-1259548185
8th edition
Authors: David Spiceland, James Sepe, Mark Nelson, Wayne Thomas
Posted Date:
Students also viewed these finance questions
-
Calculate the average PopRate for each Brand and each Time. Calculate the overall average PopRate.
-
You can use any angular measureradians, degrees, or revolutionsin some of the equations in Chapter 9, but you can use only radian measure in others. Identify those for which using radians is...
-
Calculate the contribution margin for each department and income before taxes, based on thefollowing: Dept. A 1,600 square feet Dept. B 2,400 square feet $11,000 5,900 $6,200 Net Sales Cost of Goods...
-
8. Describe each of the following types of isomerism (i) geometrical isomerism (2 marks) (ii) solavate isomerism (2 marks) (iii) coordination isomerism (2 amrks)
-
The Durkin Investing Agency has been the best stock picker in the country for the past two years. Before this rise to fame occurred, the Durkin newsletter had 200 subscribers. Those subscribers beat...
-
Designate whether the supplies and equipment are indicated or not indicated for performing the skill of hand hygiene.
-
Identify three selling situations where a memorized sales presentation may be appropriate. Explain why they would be appropriate.
-
Larrys Lawn Equipment Company gives terms of 2/10, n/30. Larry has annual credit sales of $500,000 and average accounts receivable of $60,000. a. What is Larrys accounts receivable turnover? b. What...
-
The following Trial Balance was extracted from the Books of Natasha, a Sole trader, as at 31 December 2020. Capital Furniture and Fittings Purchases Sales Stock 1 January 2020 Debtors Creditors...
-
Cesar Ablang is a product manager for a Malaysian firm. His product is made in batches of 2,500 units each. In past years, his product cost was $14.50 per unit, including $8 in variable costs. The...
-
show solutions to the following questions. There are several paths to resolution for these types of errors: Run the Install-WebApplicationProxy cmdlet again to clear up configuration issues. ...
-
A fermentation conditions were optimised on a laboratory fermenter of diameter and liquid height of 15 cm. The fermenter is equipped with a 5 cm diameter Rushton turbine operated at 800 rpm. The...
-
What volume of 2.5% (m/v) KOH can be prepared from 125 mL of a 5.0% (m/v) KOH solution? O 0.0040 mL O 125 mL 0.10 mL O 63 mL O 250 mL 4
-
What volume (in mL) of 0.350 M HCI would be required to completely react with 5.60 g of Al in the following chemical reaction? 2 Al(s) + 6 HCl(aq) 2 AICI3 (aq) + 3 H(g)
-
Which element has no stable isotopes? (4) 82Pb (1) 27 Co (2) 51Sb (3) 90Th
-
The following chemical reaction is classified as a answer may be possible. NH4NO3 (s) N (g) + 2 HO(g) + O(g) reaction. Note: more than one
-
Calculate the internal rate of return for the two following projects: Year Project A Project B 0 $-4970 $-3140 1 1710 1190 2 2980 1420 3 2130 1770 What is the IRR of project A? (Report answer in...
-
Heineken N.V., a global brewer based in the Netherlands, reports the following balance sheet accounts for the year ended December 31, 2016 (euros in millions). Prepare the balance sheet for this...
-
Access the FASB Accounting Standards Codification at the FASB website (asc.fash.org). Determine the specific citation for each of the following items: 1. What is the balance sheet classification for...
-
Creative Computing sells a tablet computer called the Protab. The $780 sales price of a Protab Package includes the following: ¢ One Protab computer. ¢ A 6-month limited warranty. This...
-
For each of the following situations, (1) indicate whether you agree or disagree with the financial reporting practice employed and (2) state the accounting concept that is applied (if agree), or...
-
If you know the electric field \(\vec{E}\) at some location, how can you determine the magnitude and direction of the electric force exerted by that field on an object carrying a charge \(q\) and...
-
(a) If the particle in Figure 23.6 carries a negative charge \(q (b) Does the electric field created by the particle point toward or away from the particle? (c) If \(q\) and \(q_{2}\) are negative,...
-
(a) If the charge on particle 2 in Exercise 23.1 is doubled so that \(q_{2}=2 q_{1}\), what happens to the direction of the electric field at points \(P_{1}\) through \(P_{4}\) ? (b) If the charge on...

Study smarter with the SolutionInn App


