Question: b. How many neutrons does a phosphorus atom have? To upload multiple files, be sure to select all of them after clicking on Browse.
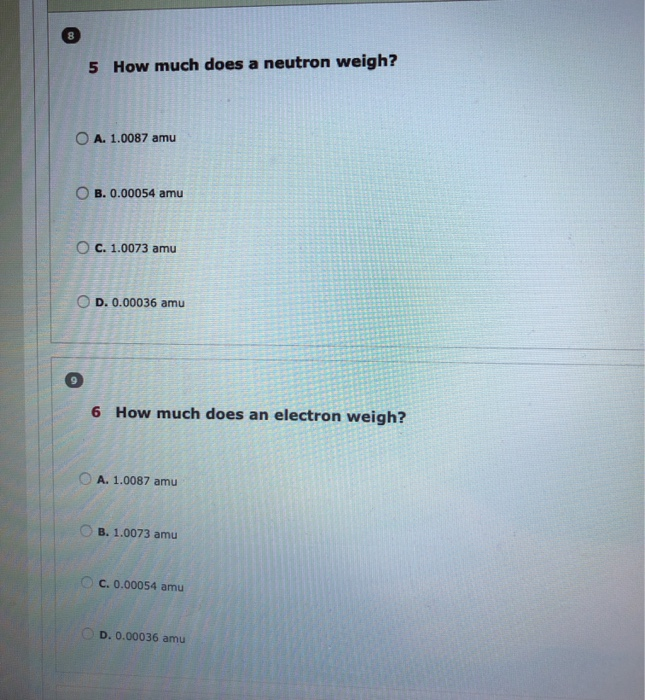
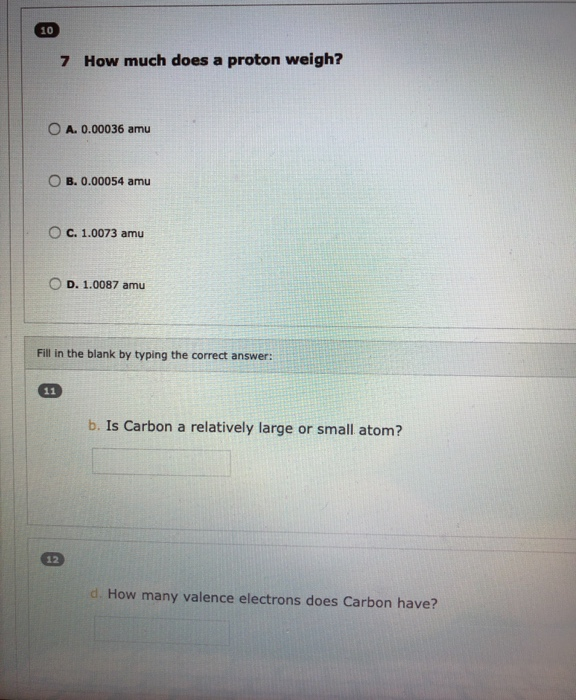
b. How many neutrons does a phosphorus atom have? To upload multiple files, be sure to select all of them after clicking on Browse. Maximum file size is 5 MB, supported file formats: txt, doc, docx, ppt, pptx, xls, xlsx, rtf, pdf, pr c. Draw and label a phosphorus atom (P) Upload your drawing below. 5 How much does a neutron weigh? O A. 1.0087 amu B. 0.00054 amu O C. 1.0073 amu OD. 0.00036 amu 6 How much does an electron weigh? OA. 1.0087 amu O B. 1.0073 amu C. 0.00054 amu D. 0.00036 amu 10 7 How much does a proton weigh? OB. 0.00054 amu A. 0.00036 amu OC. 1.0073 amu 11 12 Fill in the blank by typing the correct answer: D. 1.0087 amu b. Is Carbon a relatively large or small atom? d. How many valence electrons does Carbon have?
Step by Step Solution
3.53 Rating (173 Votes )
There are 3 Steps involved in it
Structure of Phosphorous atom Mass of neutron 1008 amu Mass of ... View full answer

Get step-by-step solutions from verified subject matter experts


