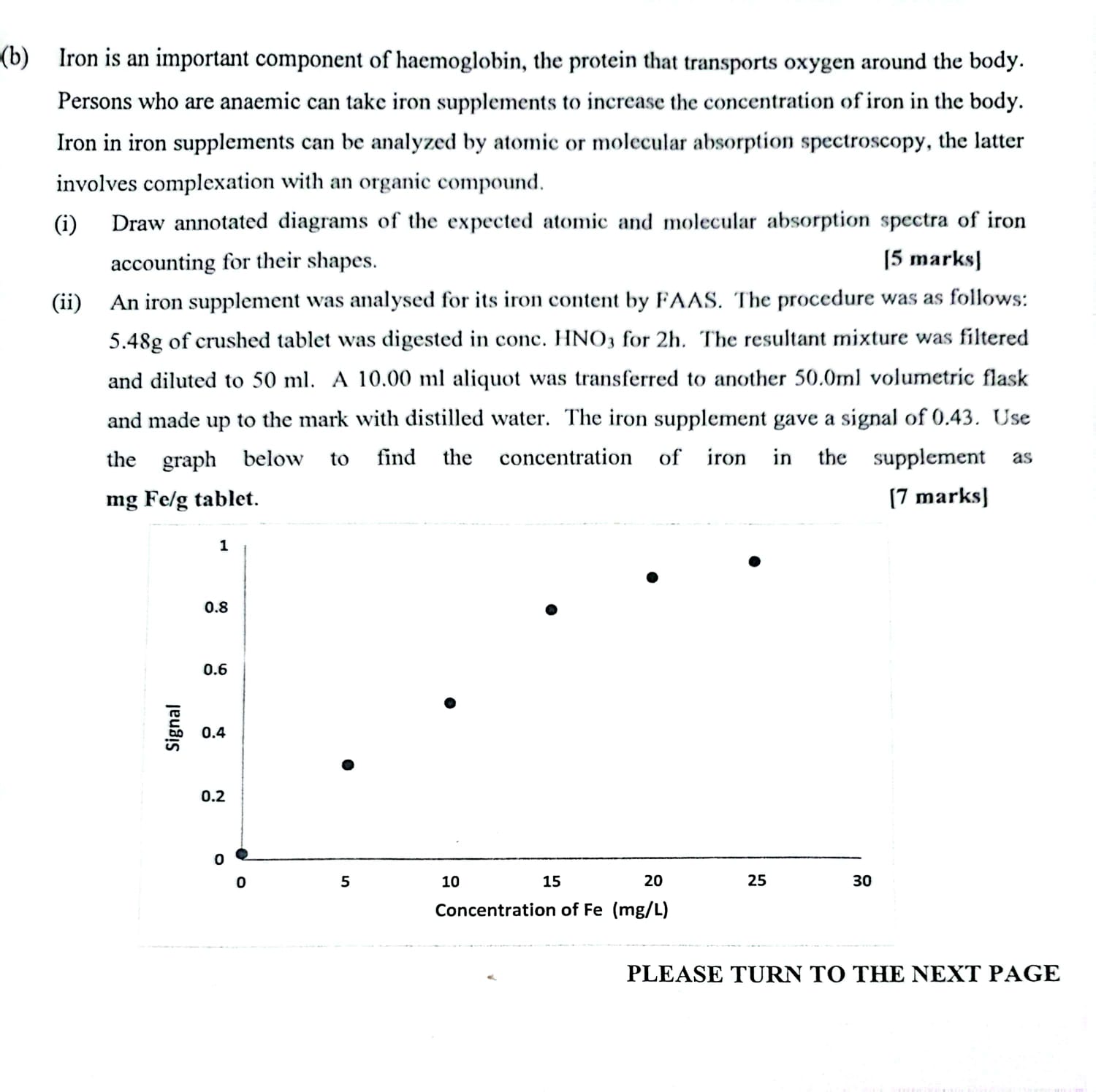
(b) Iron is an important component of haemoglobin, the protein that transports oxygen around the body....
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
(b) Iron is an important component of haemoglobin, the protein that transports oxygen around the body. Persons who are anaemic can take iron supplements to increase the concentration of iron in the body. Iron in iron supplements can be analyzed by atomic or molecular absorption spectroscopy, the latter involves complexation with an organic compound. (i) Draw annotated diagrams of the expected atomic and molecular absorption spectra of iron accounting for their shapes. [5 marks] (ii) An iron supplement was analysed for its iron content by FAAS. The procedure was as follows: 5.48g of crushed tablet was digested in conc. HNO3 for 2h. The resultant mixture was filtered and diluted to 50 ml. A 10.00 ml aliquot was transferred to another 50.0ml volumetric flask and made up to the mark with distilled water. The iron supplement gave a signal of 0.43. Use the graph below to find the concentration of iron in the supplement as mg Felg tablet. [7 marks] Signal 1 0.8 0.6 0.4 0.2 5 20 15 Concentration of Fe (mg/L) 10 25 30 PLEASE TURN TO THE NEXT PAGE (b) Iron is an important component of haemoglobin, the protein that transports oxygen around the body. Persons who are anaemic can take iron supplements to increase the concentration of iron in the body. Iron in iron supplements can be analyzed by atomic or molecular absorption spectroscopy, the latter involves complexation with an organic compound. (i) Draw annotated diagrams of the expected atomic and molecular absorption spectra of iron accounting for their shapes. [5 marks] (ii) An iron supplement was analysed for its iron content by FAAS. The procedure was as follows: 5.48g of crushed tablet was digested in conc. HNO3 for 2h. The resultant mixture was filtered and diluted to 50 ml. A 10.00 ml aliquot was transferred to another 50.0ml volumetric flask and made up to the mark with distilled water. The iron supplement gave a signal of 0.43. Use the graph below to find the concentration of iron in the supplement as mg Felg tablet. [7 marks] Signal 1 0.8 0.6 0.4 0.2 5 20 15 Concentration of Fe (mg/L) 10 25 30 PLEASE TURN TO THE NEXT PAGE (b) Iron is an important component of haemoglobin, the protein that transports oxygen around the body. Persons who are anaemic can take iron supplements to increase the concentration of iron in the body. Iron in iron supplements can be analyzed by atomic or molecular absorption spectroscopy, the latter involves complexation with an organic compound. (i) Draw annotated diagrams of the expected atomic and molecular absorption spectra of iron accounting for their shapes. [5 marks] (ii) An iron supplement was analysed for its iron content by FAAS. The procedure was as follows: 5.48g of crushed tablet was digested in conc. HNO3 for 2h. The resultant mixture was filtered and diluted to 50 ml. A 10.00 ml aliquot was transferred to another 50.0ml volumetric flask and made up to the mark with distilled water. The iron supplement gave a signal of 0.43. Use the graph below to find the concentration of iron in the supplement as mg Felg tablet. [7 marks] Signal 1 0.8 0.6 0.4 0.2 5 20 15 Concentration of Fe (mg/L) 10 25 30 PLEASE TURN TO THE NEXT PAGE
Expert Answer:
Answer rating: 100% (QA)
Solution i Atomic and molecular absorption spectra ... View the full answer

Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Posted Date:
Students also viewed these chemical engineering questions
-
Hyten Corporation On June 5, 1998, a meeting was held at Hyten Corporation, between Bill Knapp, Director of Marketing/Sales, and John Rich, director of engineering. The purpose of the meeting was to...
-
What is the effect of cultural commoditization and transformation on local traditions and customs? Give an example. Check Chapter 4 in the textbook for information on cultural commoditization and...
-
Regulation imperial soccer fields have a length of 91 meters and a width of 39 meters. Show that during a friendly match there are always two players that are not farther from each other than 19...
-
Wexpro, Inc., produces several products from processing 1 ton of clypton, a rare mineral. Material and processing costs total $60,000 per ton, one-fourth of which is allocated to product X15. Seven...
-
Write a brief verbal description of the type of linear programming problem that can be solved by the method indicated in Problem. Include the type of optimization, the number of variables, the type...
-
Write a short note on : Types of belt drive.11
-
Analyzing the Effects of Transactions in T-Accounts Lisa Frees and Amelia Ellinger had been operating a catering business for several years. In March 2011, the partners were planning to expand by...
-
Oscillations of a carbon monoxide (CO) molecule can be modeled with a harmonic oscillator. What is the size of energy of the harmonic oscillator at ground state when a CO molecule absorbs radiation...
-
Reconsider the experiment described in Problem 6-1. Suppose that the experimenter only performed the eight trials from replicate I. In addition, he ran four center points and obtained the following...
-
11. Identify the following drugs and provide an explanation for their mechanism of action. HN HDHC
-
In assessing/evaluating the nine enablers to collaboration, identify and explain those enablers that, in your experience, have proven to be the most important/critical to successful collaboration....
-
a) If x t logt and y= log find dy at t=1. dx b) Differentiate sinx with respect to logx.
-
Write the number in standard form. 16--192 4
-
On December 1, Jasmin Ernst organized Ernst Consulting. On December 3, the owner contributed $84,780 in assets in exchange for its common stock to launch the business. On December 31, the company's...
-
2) Assume that your widget manufacturing company has a total annual demand of N widgets per year evenly distributed across the year. Each widget cost $b dollars in material and manufacturing costs to...
-
A trust that is required to distribute all of its income annually receives a exemption for the year of A) $0, because it retains no income. B) $100. C) $300. D) $600.
-
Reconsider Prob. 1474. In order to drain the tank faster, a pump is installed near the tank exit as in Fig. P1475. Determine how much pump power input is necessary to establish an average water...
-
Calculate the pH of each of the following solutions: (a) 2.8 x 10-4 M Ba(OH)2, (b) 5.2 x 10-4 M HNO3.
-
In Breckenridge, Colorado, the typical atmospheric pressure is 520 torr. What is the boiling point of water (Hvap = 40.7 kJ/ mol) in Breckenridge? What pressure would have to be applied to steam at...
-
Consider the following galvanic cell: Calculate the concentrations of Ag+(aq) and Ni2+(aq) once the cell is dead. Ni 1.0 MNi2+ 10 M Ag
-
What are the stages in the product life cycle?
-
What is the hype cycle?
-
What is meant by the term agile innovation?

Study smarter with the SolutionInn App


