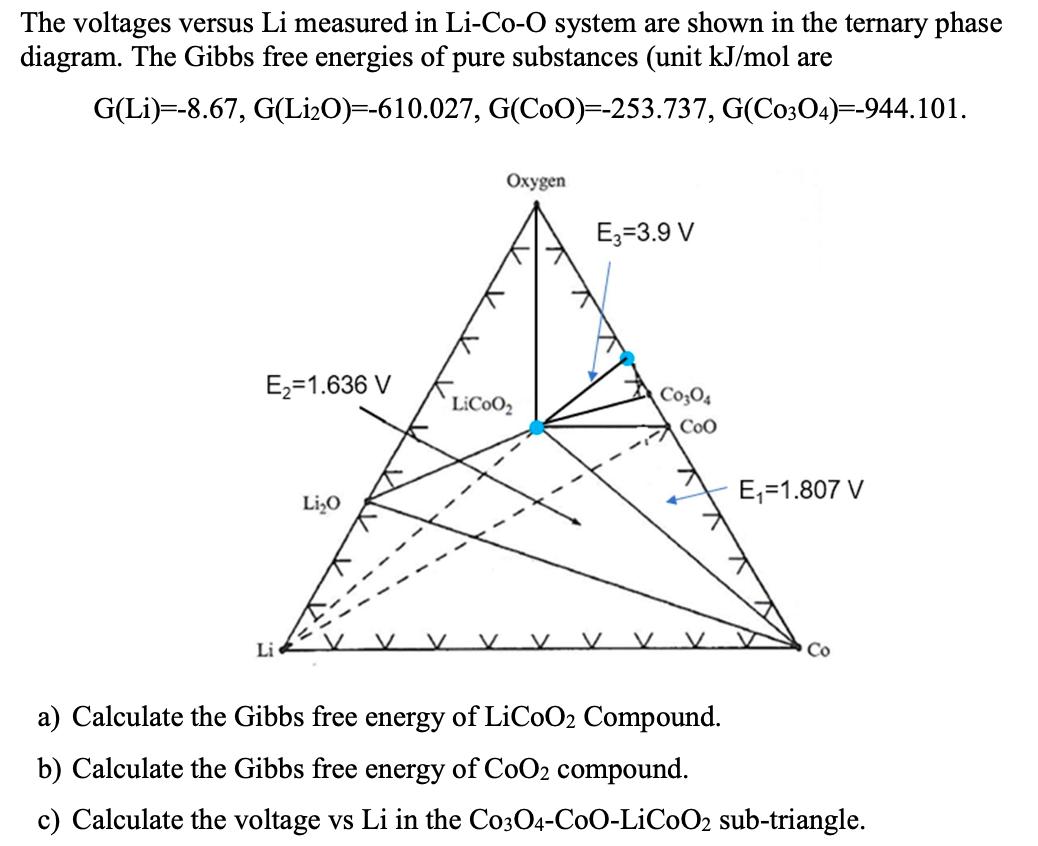
The voltages versus Li measured in Li-Co-O system are shown in the ternary phase diagram. The...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
The voltages versus Li measured in Li-Co-O system are shown in the ternary phase diagram. The Gibbs free energies of pure substances (unit kJ/mol are G(Li) -8.67, G(Li₂O)=-610.027, G(COO)=-253.737, G(C0304)=-944.101. E₂=1.636 V Li Li₂O Oxygen LiCoo₂ E3-3.9 V V C0304 Coo E₁=1.807 V Co a) Calculate the Gibbs free energy of LiCoO2 Compound. b) Calculate the Gibbs free energy of CoO2 compound. c) Calculate the voltage vs Li in the C0304-C0O-LiC0O2 sub-triangle. The voltages versus Li measured in Li-Co-O system are shown in the ternary phase diagram. The Gibbs free energies of pure substances (unit kJ/mol are G(Li) -8.67, G(Li₂O)=-610.027, G(COO)=-253.737, G(C0304)=-944.101. E₂=1.636 V Li Li₂O Oxygen LiCoo₂ E3-3.9 V V C0304 Coo E₁=1.807 V Co a) Calculate the Gibbs free energy of LiCoO2 Compound. b) Calculate the Gibbs free energy of CoO2 compound. c) Calculate the voltage vs Li in the C0304-C0O-LiC0O2 sub-triangle.
Expert Answer:
Answer rating: 100% (QA)
Well use the relationship between Gibbs free energy and this problem to solve it Voltage VE and temp... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
A physician prescribes an ophthalmic suspension to contain 100 mg of cortisone acetate in 8 mL of normal saline solution. The pharmacist has on hand a 2.5% w/v suspension of cortisone acetate in...
-
In the Continuing Payroll Problem A, presented at the end of succeeding chapters, you will gain experience in computing wages and salaries and preparing a payroll register for Kipley Company, Inc., a...
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
- Given the following data for a company: Equity E 800,000 USD -Debt D=200,000 USD - Invested Capital IC = 1,000,000 USD E/IC-80% - D/IC=20% -Cost of debt k=9% - Beta-1.6 - D/E=0.25 compute the cost...
-
Two forces P and Q are applied as shown to an aircraft connection. Knowing that the connection is in equilibrium and that P = 500lb and Q= 650lb, determine the magnitudes of the forces exerted on the...
-
Simulate drawing 100 simple random samples of size n = 15 from a population that is normally distributed with mean 100 and standard deviation 15. (a) Test the null hypothesis H0: = 100 versus H1: ...
-
Kokomochi is considering the launch of an advertising campaign for its latest dessert product, the Mini Mochi Munch. Kokomochi plans to spend \($4.78\) million on TV, radio, and print advertising...
-
1. Draw a Gantt chart for the construction phase of the program. What is the completion date if construction starts in March? What is the completion date of the project if construction is started in...
-
Mrs. J has a history of pernicious anemia and visits her physicians office bi-monthly for vitamin B12 injections. Order: vitamin B12 0.50 mg IM bi-monthly. Supply: vitamin B12 1000mcg/mL. How many mL...
-
The following selected accounts and their current balances appear in the ledger of Druid Hills Co. for the fiscal year ended May 31, 20Y8: Cash .......................................... $ 290,800...
-
The Angla Ruston University operates a Laundromat, solely for the purpose of providing laundering services to students in residence. You have been hired as accounting students to analyze the cost of...
-
A +4.0-C charge is placed on the x axis at x = +3.0 m, and a -2.0-C charge is located on the y axis at y = -1.0 m. Point A is on the y axis at y = +4.0 m. Determine the electric potential at point A...
-
How do you check for accounts with a UID of 0 at the terminal?
-
A 0.800 kg hammer is moving horizontally at 9.50 m/s when it strikes a nail and comes to rest after driving it 1.00 cm into a board. A. Calculate the duration of the impact in seconds. B. What was...
-
Which lines of the code below represents the base case of the recursion? def tree op (fn, t): #1 #2 #3 if type (t) !=type ([]): return t left = tree op (fn, t[0]) right tree op (fn, t[1]) return fn...
-
a. Explain how you can tell if an expression is a tautology, a contradiction, or neither? b. Make a truth table for ALL possible cases of this logical expression: P^(-(QVR)) c. Is it a tautology, a...
-
Define Matrix Arithmetic and give 2 examples.
-
suppose a nickel-contaminated soil 15 cm deep contained 800 mg/kg Ni, Vegetation was planted to remove the nickel by phytoremediation. The above-ground plant parts average 1% Ni on a dry-weight bas...
-
The ethylene epoxydation is to be carried out using a cesium-doped silver catalyst in a packed-bed reactor. Along with the desired reaction, the complete combustion of ethylene also occurs It is...
-
A gas-phase reaction is being carried out in a 5-cm-diameter tubular reactor that is 2 m in length. The velocity inside the pipe is 2 cm/s. As a very first approximation, the gas properties can be...
-
The relative tracer concentrations obtained from pulse tracer tests on a commercial packed-bed desulfurization reactor are shown in Figure P17-18B. After studying the RTD, what problems are occurring...
-
Using Mendels data from the experiment in Figure 2.8, conduct a chi square test to determine if the data agree with Mendels law of independent assortment. Mendels data: THE DATA P cross F1 generation...
-
How long did it take Mendel to complete the experiment in Figure 2.5? Figure 2.5 Experimental level P plants Tall Dwarf Note: The P cross produces seeds that are part of the Fi generation. Tall Self-...
-
On rare occasions, an organism may have three copies of a chromosome and therefore three copies of the genes on that chromosome (instead of the usual number of two copies). The alleles for each gene...

Study smarter with the SolutionInn App


