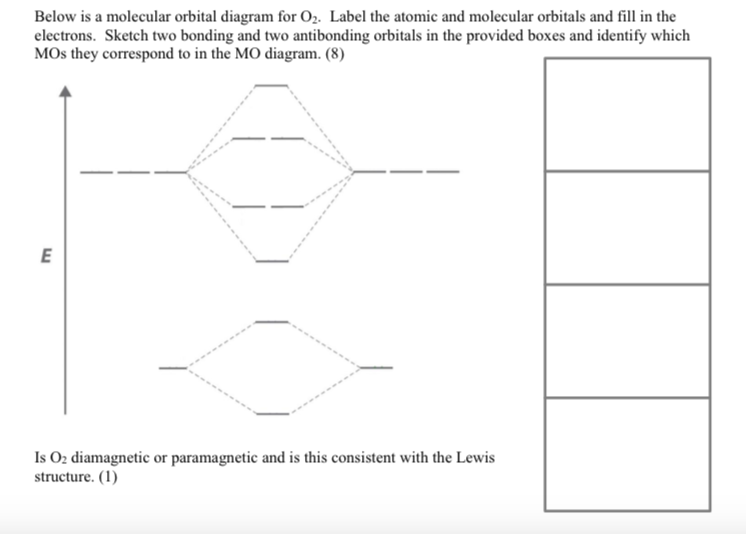
Below is a molecular orbital diagram for O. Label the atomic and molecular orbitals and fill...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Below is a molecular orbital diagram for O₂. Label the atomic and molecular orbitals and fill in the electrons. Sketch two bonding and two antibonding orbitals in the provided boxes and identify which MOs they correspond to in the MO diagram. (8) E Is O₂ diamagnetic or paramagnetic and is this consistent with the Lewis structure. (1) Below is a molecular orbital diagram for O₂. Label the atomic and molecular orbitals and fill in the electrons. Sketch two bonding and two antibonding orbitals in the provided boxes and identify which MOs they correspond to in the MO diagram. (8) E Is O₂ diamagnetic or paramagnetic and is this consistent with the Lewis structure. (1)
Expert Answer:
Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
i. Construct Molecular Orbital diagram for C 2 -2 ii. Write the electron configuration for C 2 -2 iii. Identify the magnetism for C 2 -2 iv. State the HOMO and LUMO for C 2 -2 v. Calculate the bond...
-
Draw the complete molecular orbital diagram for Dichlorobis(ethylenediamine)cobalt(III), considering only the -bonding.
-
Sketch the molecular orbital and label its type (Ï or p; bonding or antibonding) that would be formed when the following atomic orbitals overlap. Explain your labels. a. b. d.
-
What is opportunity cost? explain a time in your life when you experienced opportunity cost. Why did you make the decision? What was another alternative that you could have made? Discuss how scarcity...
-
Harley worked for many years to save enough money to start his own residential landscape design business. The cash flows shown are those he recorded for the first 6 years as his own boss. Find the...
-
Answer the following questions: a. Can a routing table in a datagram network have two entries with the same destination address? Explain. b. Can a switching table in a virtual-circuit network have...
-
Your business purchased supplies of \($1,000\) on account. The journal entry to record this transaction is: a. Supplies 1,000 Accounts receivable 1,000 b. Accounts payable 1,000 Supplies 1,000
-
Using Table 8.4, select possible liquid liquid extraction solvents for separating the following mixtures: (a) Waterethyl alcohol, (b) Wateraniline, and (c) Wateracetic acid. For each case, indicate...
-
= I. Let P (-2.3). Q = (3.13), R=P+Q. v=Q-P and P+[e]. Moreover, let X=(1, 2) be any point of E. 1. Find (P.Q). 2. Find v. 3. Write the equation of the line in the form ar+by+c=0. 4. Find A and a...
-
Wedding Planners Limited (WP), owned by Anne and Francois Tremblay, provides wedding planning and related services. WP owns a building (the Pavilion) that has been custom-made for hosting weddings....
-
Critically appraise the purposes and goals of activities within project management context give examples.
-
Imagine a cannonball being launched from a cannon atop a very high cliff. Imagine as well that the cannonball does not encounter a significant amount of air resistance. What will be the path of the...
-
On 8 February 19X8 PJ, a London trader, consigned 120 cases of goods to MB, an agent in New Zealand. The cost of the goods was 25 a case. PJ paid carriage to the port 147 and insurance 93. On 31...
-
01.As shown below, an object is moving uniformly from location A to location "B' as specified by their co-ordinates. 1011 ay 3 A(2,4) at time(t)- 5 Secs 6 2 3 4 5 6 7 (a) Determine the distance...
-
A bond is currently selling in the market for $1,085.96. It has a coupon of 8% and a 15-year maturity. Using annual compounding, what is the yield to maturity on this bond?
-
A block of mass=315g is dragged with a string across a rough horizontal table. The string tension is=2.97,and it pulls upward at an angle of 46.0 with the horizontal. At one particular instant, the...
-
5. The economies of New Zealand and Australia produce only wheat and cotton. Each country has land area of 100 acres. The following table gives the production per acre of what and Cotton in each...
-
Write the expression in radical notation. Then evaluate the expression when the result is an integer. 23 -1/2
-
Account for the following observations: (a) H3PO3 is a diprotic acid. (b) Nitric acid is a strong acid, whereas phosphoric acid is weak. (c) Phosphate rock is ineffective as a phosphate fertilizer....
-
The osmotic pressure of a saturated solution of strontium sulfate at 25c is 21 torr. What is the solubility product of this salt at25?
-
Ortho-dichlorobenzene, C6H4Cl2, is obtained when two of the adjacent hydrogen atoms in benzene are replaced with Cl atoms. A skeleton of the molecule is shown here. (a) Complete a Lewis structure for...
-
A reversible process is a process (a) Which proceeds with no driving force (b) Which takes place spontaneously (c) Which is quasi-static (d) Which is frictional process.
-
At constant temperature and pressure, the free energy for a chemically reacting system at equilibrium is (a) Minimum (b) Maximum (c) Can not be predicted (d) None of these.
-
The operation of a throttling device follows the (a) Zeroth law of thermodynamics (b) First law of thermodynamics (c) Second law of thermodynamics (d) Third law of thermodynamics.

Study smarter with the SolutionInn App



