Question: C2021 HW Part 1 + Isothermal Engine + Isothermal Engine Consider a thermal engine filled with N molecules of a monatomic ideal gas. The
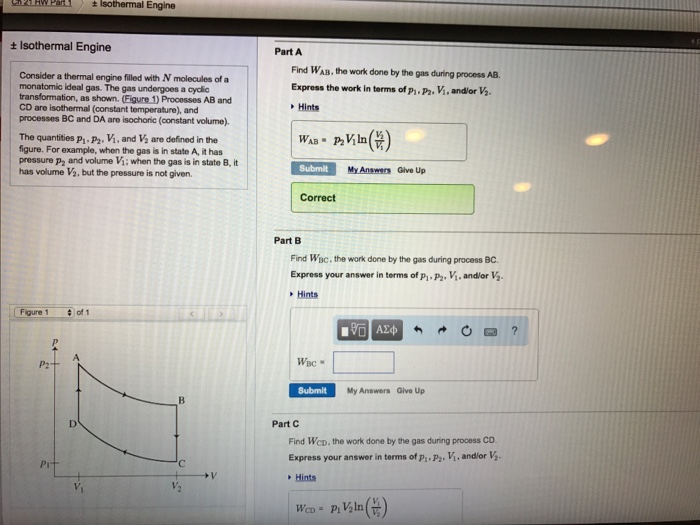
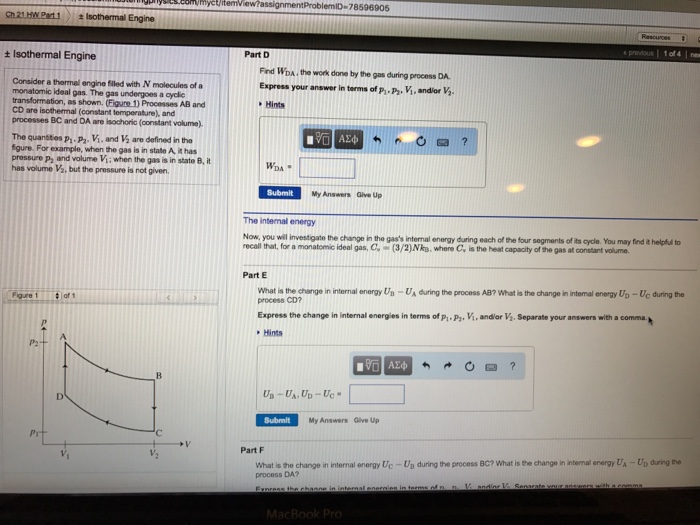
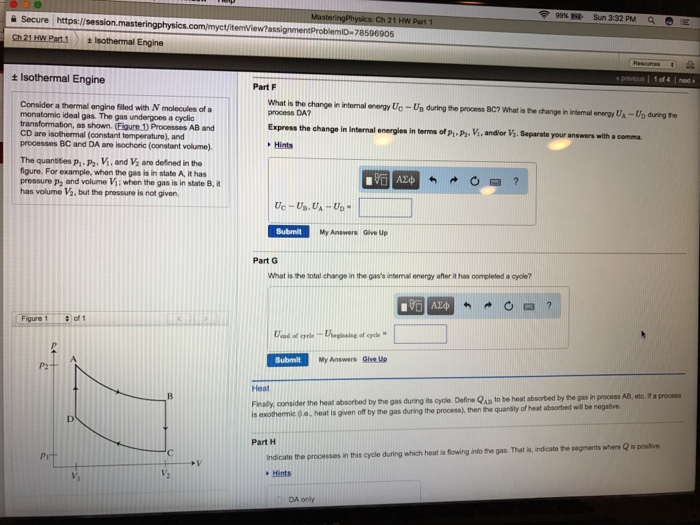
C2021 HW Part 1 + Isothermal Engine + Isothermal Engine Consider a thermal engine filled with N molecules of a monatomic ideal gas. The gas undergoes a cyclic transformation, as shown. (Figure 1) Processes AB and CD are isothermal (constant temperature), and processes BC and DA are isochoric (constant volume). The quantities P, P. V, and V are defined in the figure. For example, when the gas is in state A, it has pressure p and volume V; when the gas is in state B, it has volume V, but the pressure is not given. Figure 1 P2 Pr of 1 ' V Part A Find WAB, the work done by the gas during process AB. Express the work in terms of P. P2. V, and/or V. Hints WAB- P. Viln (V) Submit Correct Part B Find Wac, the work done by the gas during process BC. Express your answer in terms of p. P, V, and/or V. Hints WBC My Answers Give Up Submit 195| My Answers Give Up Part C Find Wcp, the work done by the gas during process CD. Express your answer in terms of p, P, V, and/or V. Hints WCD-P Vln() ? Ch 21 HW Part 1 + Isothermal Engine Consider a thermal engine filled with N molecules of a monatomic ideal gas. The gas undergoes a cyclic transformation, as shown. (Figure 1) Processes AB and CD are isothermal (constant temperature), and processes BC and DA are isochoric (constant volume). Figure 1 The quantities P P. V. and V are defined in the figure. For example, when the gas is in state A, it has pressure p, and volume V; when the gas is in state B, it has volume V, but the pressure is not given. 2 Pit Isothermal Engine of 1 D myct/itemView?assignment ProblemiD-78596905 'C Part D Find WDA, the work done by the gas during process DA Express your answer in terms of P.P, V, and/or V. Hints WDA 195] Submit My Answers Give Up The internal energy Now, you will investigate the change in the gas's internal energy during each of the four segments of its cycle. You may find it helpful to recall that, for a monatomic ideal gas, C, (3/2) Nks, where C, is the heat capacity of the gas at constant volume. Part E What is the change in internal energy UB-UA during the process AB? What is the change in internal energy Up-Uc during the process CD? Express the change in internal energies in terms of P. P, V, and/or V. Separate your answers with a comma.p Hints UB-UA. UD-Uc 197| Submit My Answers Give Up Resources # previous 1 of 4 new MacBook Pro ? Part F What is the change in internal energy Uc-Us during the process BC? What is the change in internal energy UA-Up during the process DA? Express the change in internal energies in terms of n n Vanding V. Senarate your answers with a comma Secure Ch.21 HW Part.1 + Isothermal Engine Consider a thermal engine filled with N molecules of a monatomic ideal gas. The gas undergoes a cyclic transformation, as shown. (Figure 1) Processes AB and CD are isothermal (constant temperature), and processes BC and DA are isochoric (constant volume). Figure 1 The quantities p-P. V, and V are defined in the figure. For example, when the gas is in state A, it has pressure p, and volume V; when the gas is in state B, it has volume V, but the pressure is not given. P2 https://session.masteringphysics.com/myct/itemView?assignment 24 Pit Isothermal Engine of 1 V B 'C V ProblemiD-78596905 MasteringPhysics: Ch 21 HW Part 1 +V Uc-UB. UA-UD" Submit My Answers Give Up Part F What is the change in internal energy Ue-Us during the process BC? What is the change in intermal energy UA-Up during the process DA? Express the change in internal energies in terms of P-P. V, and/or V. Separate your answers with a comma. Hints Part G What is the total change in the gas's internal energy after it has completed a cycle? Uend of cycle - Ubeginning of cycle Submit My Answers Give Up DA only 99% ? Sun 3:32 PM q 1 previous 1 of 4 | next H Heat Finally, consider the heat absorbed by the gas during its cycle. Define Qs to be heat absorbed by the gas in process AB, etc. If a process is exothermic (e. heat is given off by the gas during the process), then the quantity of heat absorbed will be negative. Part H Indicate the processes in this cycle during which heat is flowing into the gas. That is, indicate the segments where is positive Hints
Step by Step Solution
3.43 Rating (172 Votes )
There are 3 Steps involved in it
Part B The work done during the gas along BC would be equal to W BC PV final V initial As it is an i... View full answer

Get step-by-step solutions from verified subject matter experts


