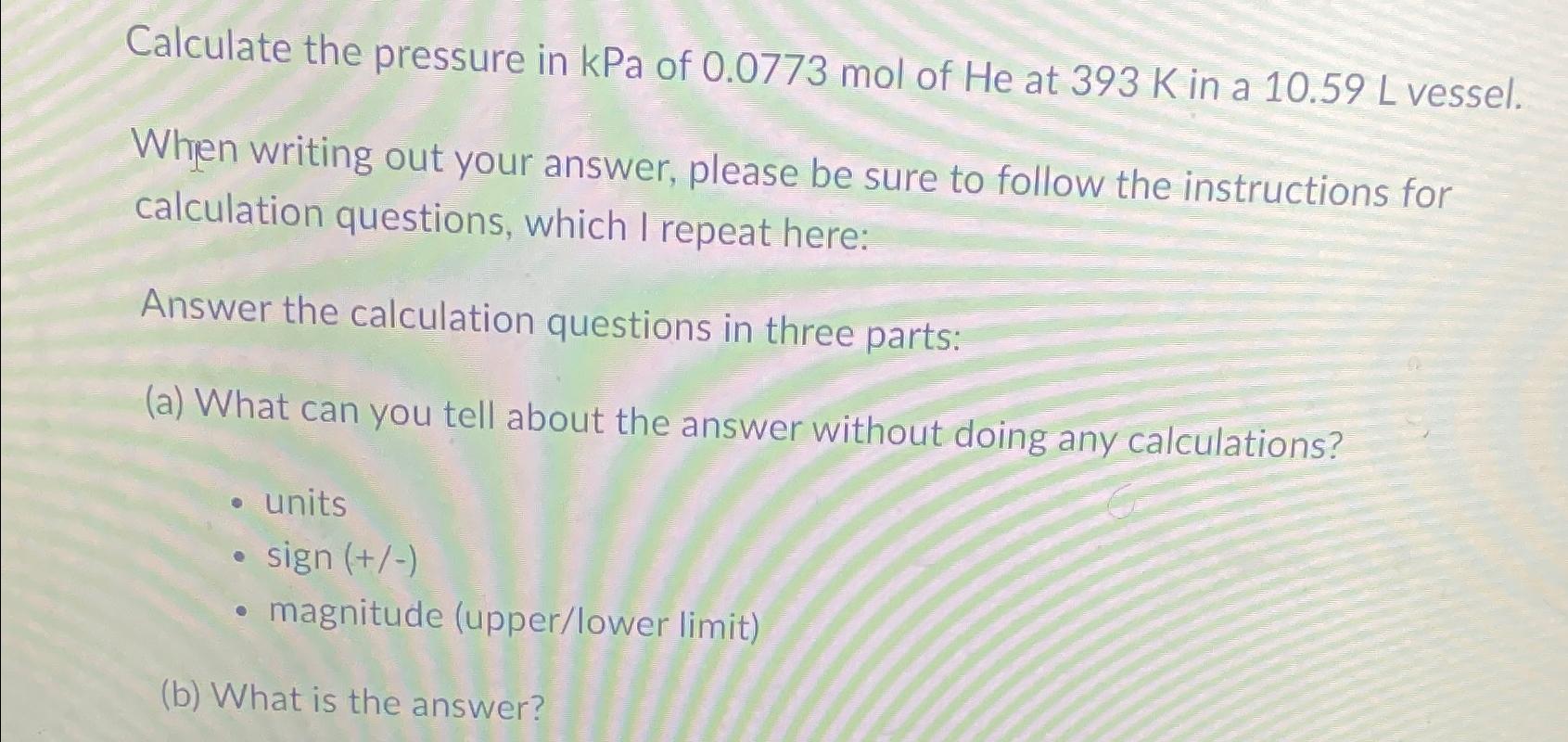
Calculate the pressure in kPa of 0.0773 mol of He at 393 K in a 10.59...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Calculate the pressure in kPa of 0.0773 mol of He at 393 K in a 10.59 L vessel. When writing out your answer, please be sure to follow the instructions for calculation questions, which I repeat here: Answer the calculation questions in three parts: (a) What can you tell about the answer without doing any calculations? units sign (+/-) magnitude (upper/lower limit) (b) What is the answer? Calculate the pressure in kPa of 0.0773 mol of He at 393 K in a 10.59 L vessel. When writing out your answer, please be sure to follow the instructions for calculation questions, which I repeat here: Answer the calculation questions in three parts: (a) What can you tell about the answer without doing any calculations? units sign (+/-) magnitude (upper/lower limit) (b) What is the answer?
Expert Answer:
Answer rating: 100% (QA)
The image you provided displays a question that asks for the calculation of pressure in kilopascals ... View the full answer

Related Book For 

Managerial accounting
ISBN: 978-0471467854
1st edition
Authors: ramji balakrishnan, k. s i varamakrishnan, Geoffrey b. sprin
Posted Date:
Students also viewed these chemical engineering questions
-
Q1) The shape given in the figure below is located on Frame A with an offset of 2m on ya-axis and the circle M is located on the middle of the surface of the shape. Zc X Yc+ C Za 5m A Xc Va 2m 1m Xb...
-
Suppose a U.S. manufacturer, Patton-Stillwell (P-S, Inc), sells its merchandise on account to a firm in the Netherlands for 1,800,000 (euro) expecting payment to be made in 60-days. The spot rate is...
-
Biological assets within the scope of IAS 41 are measured on initial recognition and at subsequent reporting dates at fair value less estimated costs to sell, unless fair value cannot be reliably...
-
How many moles of carbon dioxide, CO2, are in a 22 gram sample of the compound?
-
For the frame and loading shown, draw the free-body diagram(s) needed to determine the force in member BD and the components of the reaction at C. 135 mm 240 min 450 mm 510 mm 120 mma 400 N
-
Explain the relationship between inventory turnover and purchasing needs.
-
A red \(10-\mathrm{kg}\) cart is connected to a \(20-\mathrm{kg}\) cart by a relaxed spring of spring constant \(60 \mathrm{~N} / \mathrm{m}\). The \(20-\mathrm{kg}\) cart is resting against another...
-
In 2013, Manhoff Company had a break-even point of $350,000 based on a selling price of $5 per unit and fixed costs of $112,000. In 2014, the selling price and the variable costs per unit did not...
-
Selected information from the lowa Instruments accounting records for April follows: Materials Inventory Work-in-Process Inventory Credit Debit Credit BB (4/1) 27,400 211,400 Debit Labor 97,200...
-
Ryan Ross (111-11-1112), Oscar Omega (222-22-2222), Clark Carey (333-33-3333), and Kim Kardigan (444-44-4444) are equal active members in ROCK the Ages LLC. ROCK serves as agents and managers for...
-
Business Process Management (BPM) is an approach to analysing and optimising business processes. While the approach is criticised for its inability to deal with uncertainty and continuous and...
-
A proton starts out at the central point of a cyclotron with radius R = 5 0 cm in which a uniform magnetic field B = 1 T is established perpendicular to the plane of motion of the proton. Determine:...
-
Suppose object A is electrically charged and is experiencing forces from three other charged objects. How should the total effects of those three objects be combined in order to find the total...
-
4. How would you find the resistance of a parallel circuit with n identical resistors? (1 mark) I 5. What will be the total resistance and current in a parallel circuit with a 15-volt battery and...
-
Vector A is 5 . 2 units long at a 5 1 0 angle with respect to the positive x - axis. Vector B is 1 5 . 1 units long at an angle of 2 0 5 0 . Determine the difference between the vectors, C = A - B in...
-
The average person passes out at an acceleration of 7 g ( that is , seven times the gravitational acceleration on Earth ) . Suppose a car is designed to accelerate at this rate. How much time would...
-
What are models of solar cell and photovoltaic?
-
Assume that a trial balance is prepared with an account balance of $21,360 listed as $21,630 and an account balance of $1,500 listed as $15,000. Identify the transposition and the slide.
-
Jackrabbit Trails is a family camp located in Kings Canyon National Park, California. Open from mid-May to Labor Day, Jackrabbit Trails offers week-long family summer vacation packages that include...
-
Suppose we are choosing between two drivers to allocate the costs in the perform setup cost pool: the number of setups or the number of setup hours. When will the choice not matter (i.e., will result...
-
Research the term Modified Accelerated Cost Recovery System (MACRS) and explain its relevance to project evaluation.
-
Who was Phar-Mors flamboyant Chief Executive Officer?
-
Which of the following generally is not considered something of value? 1. Cash, money or checks 2. Airline miles or hotel credits associated with frequent activity (e.g., frequent flier miles) 3. An...
-
Which of the following is not one of the five major categories of fraudulent disbursements? 1. Payroll schemes 2. Expense reimbursement schemes 3. Shell company schemes 4. Billing schemes

Study smarter with the SolutionInn App


