III. Preparing HCH30 Solutions and Determining pH concentration of HCHO, M 1.0 x 10-1 1.0 x...
Fantastic news! We've Found the answer you've been seeking!
Question:


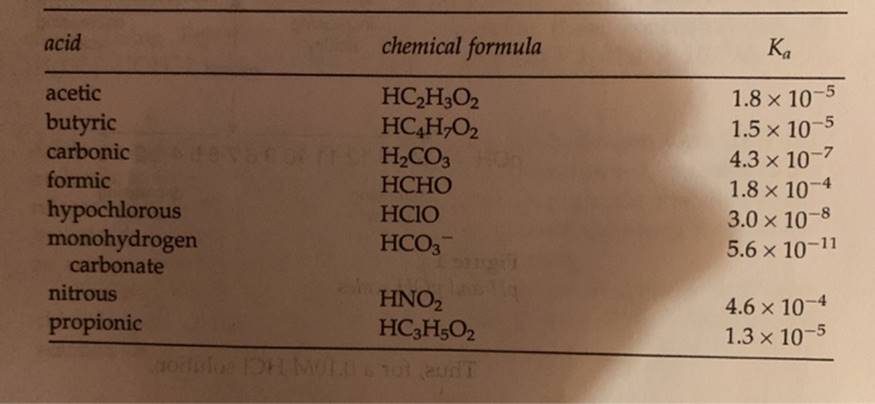
Transcribed Image Text:
III. Preparing HC₂H30₂ Solutions and Determining pH concentration of HC₂H₂O₂, M 1.0 x 10-1 1.0 x 10-2 1.0 x 10 1.0 x 104 measured pH 3.7 theoretical pH concentration of HC₂H302, M 1.0 x 10-¹ 1.0 x 10-² 1.0 × 10-3 1.0 x 104 calculated K, of HC₂H₂O2 based on pH data literature Ka of HC₂H302 acid acetic butyric carbonic formic hypochlorous monohydrogen carbonate nitrous propionic chemical formula HC₂H3O2 HC4H₂O₂ H₂CO3 HO HCHO HCIO HCO3™ HNO₂ HC3H5O2 gorilus DH MOULD6101 T Ka 1.8 x 10-5 1.5 x 10-5 4.3 x 10-7 1.8 x 10-4 3.0 × 10-8 x 5.6 × 10-11 4.6 x 10-4 1.3 × 10-5 III. Preparing HC₂H30₂ Solutions and Determining pH concentration of HC₂H₂O₂, M 1.0 x 10-1 1.0 x 10-2 1.0 x 10 1.0 x 104 measured pH 3.7 theoretical pH concentration of HC₂H302, M 1.0 x 10-¹ 1.0 x 10-² 1.0 × 10-3 1.0 x 104 calculated K, of HC₂H₂O2 based on pH data literature Ka of HC₂H302 acid acetic butyric carbonic formic hypochlorous monohydrogen carbonate nitrous propionic chemical formula HC₂H3O2 HC4H₂O₂ H₂CO3 HO HCHO HCIO HCO3™ HNO₂ HC3H5O2 gorilus DH MOULD6101 T Ka 1.8 x 10-5 1.5 x 10-5 4.3 x 10-7 1.8 x 10-4 3.0 × 10-8 x 5.6 × 10-11 4.6 x 10-4 1.3 × 10-5
Expert Answer:
Related Book For 

Cambridge International AS And A Level Chemistry Coursebook
ISBN: 9781316637739
2nd Edition
Authors: Lawrie Ryan, Roger Norris
Posted Date:
Students also viewed these accounting questions
-
3 / 4 1 1 / 4 x < 3
-
Consider the following problem. Maximize Z = -x1 + 4x2, subject to -3x1 + x2 6 x1 + 2x 4 and x2-3, (no lower bound constraint for x) Identify the optimal solution graphically
-
4 cubed + 10 X 20 + 8 squared 23
-
We often speak of how price rations goods. What are other rationing measures in clinics in which free care is provided?
-
Ace Pencil, the Web manager of the XYZ Company, was horrified when he received a call one night around midnight from Bob Drake, one of the salespersons. Our entire Web site has been replaced by...
-
As you continue to work on the chargemaster review, you must enter data for a new service that is being provided by the Radiology Department. One of the elements you need to add is the numeric value...
-
For each of the following situations, calculate the degrees of freedom \((d f)\), identify the critical values (assume \(\mathrm{a}=.05\) [two-tailed]), calculate the standard error of the mean \(s\)...
-
The Education Agency of one of the nations most populous states evaluates public elementary schools on the basis of the following inputs and outcomes: Outcomes Total campus attendance Average...
-
On January 1 5 , 2 0 2 3 , Dave Quinn, a U . S . citizen, formed a GmbH in Germany to own and operate a building in Hamburg, Germany. Briefly discuss the answers to the following questions: a ....
-
One of the shops in hackerMall is offering discount coupons based on a puzzling porblem. There are n tags, where each tag has a value denoted by val [ [ i ] . ] . A customer needs to choose teh tags...
-
(1) A part of an accountant's task is substantial involvement in setting financial targets and budgets within an organisation. Required: Describe how financial target setting can motivate employees...
-
Do you feel that it is a coach's responsibility to teach sportsmanship? Then, identify at least one coach and one professional athlete you have observed demonstrating good or bad character and...
-
(a)Describe the labor theory of value and the problems associated with the theory. (b)Are bilateral trade negotiations superior to multilateral negotiations? Include in your discussion points that...
-
1. Why do we need to calculate average time in waiting line? what kind of issues are related to average time in waiting line? 2. Why do we need to calculate Average number waiting in line?what kind...
-
How do you know if you would be a good IT project manager? After all, being an IT project manager is a demanding yet rewarding endeavor. Imagine you are chosen to be the IT project manager and must...
-
Why is it that some organizations do not place enough importance on disaster recovery? What are examples of the type of disasters that could happen? What are some things that might happen to these...
-
How might Mcdonald's adapt its business model to make it less sensitive to a similar shock in the future? Provide a brief executive summary of your suggestion. Do not quote directly from any source
-
Currently, there are five concepts of food stands, including: hot dogs, soft pretzels, turkey legs, sandwich wraps, and funnel cakes. This approach will double the existing number of food stands...
-
Suggest a suitable reagent that can carry out each of the following oxidations or reductions. Use the data in Appendix 2 to help you. a. The reduction of Zn 2+ ions to Zn. b. The oxidation of Br ...
-
An aqueous solution of silver nitrate is electrolysed. a. i. Explain why silver rather than hydrogen is produced at the cathode. ii. Write an equation for the reaction occurring at the cathode. b. i....
-
a. Calculate the volume occupied by 272 g of methane at a pressure of 250 kPa and a temperature of 54 C. (R = 8.31 J K 1 mol 1 ; M r methane = 16.0) b. The pressure exerted by 0.25 mol of carbon...
-
Sample size = 81, sample mean = 4.5 km, sample standard deviation = 3.1 km Assume that population means are to be estimated from the samples described. In each case, use the sample results to...
-
n = 100, x = 8.0 ft, s = 2.0 ft Assume that population means are to be estimated from the samples described. In each case, use the sample results to approximate the margin of error and 95% confidence...
-
Margin of error = \($5\), standard deviation = \($20\)

Study smarter with the SolutionInn App



