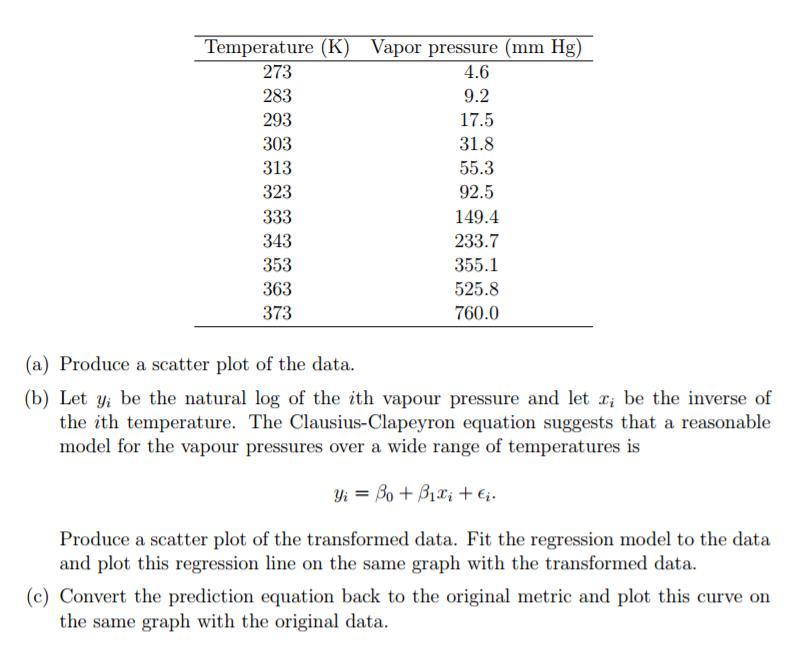
Chemical and mechanical engineers often need to know the vapour pressure of water for specific temperatures....
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
Chemical and mechanical engineers often need to know the vapour pressure of water for specific temperatures. Physical chemistry suggests that the vapour pressure should follow an exponential relationship to the inverse of the temperature. Specifically, let p, be the vapour pressure and T be the temperature, the Clausius-Clapeyron equation states that In p, is directly proportional to -1/T, that is 1 In p, ox T The following table lists the vapour pressures of water for various temperature (in Kelvin) from 0°C to 100°C. Temperature (K) Vapor pressure (mm Hg) 273 4.6 283 9.2 293 17.5 303 31.8 313 55.3 323 92.5 333 149.4 343 233.7 353 355.1 363 525.8 373 760.0 (a) Produce a scatter plot of the data. (b) Let y; be the natural log of the ith vapour pressure and let r, be the inverse of the ith temperature. The Clausius-Clapeyron equation suggests that a reasonable model for the vapour pressures over a wide range of temperatures is Yi = Bo + B1x; +6. Produce a scatter plot of the transformed data. Fit the regression model to the data and plot this regression line on the same graph with the transformed data. (c) Convert the prediction equation back to the original metric and plot this curve on the same graph with the original data. Chemical and mechanical engineers often need to know the vapour pressure of water for specific temperatures. Physical chemistry suggests that the vapour pressure should follow an exponential relationship to the inverse of the temperature. Specifically, let p, be the vapour pressure and T be the temperature, the Clausius-Clapeyron equation states that In p, is directly proportional to -1/T, that is 1 In p, ox T The following table lists the vapour pressures of water for various temperature (in Kelvin) from 0°C to 100°C. Temperature (K) Vapor pressure (mm Hg) 273 4.6 283 9.2 293 17.5 303 31.8 313 55.3 323 92.5 333 149.4 343 233.7 353 355.1 363 525.8 373 760.0 (a) Produce a scatter plot of the data. (b) Let y; be the natural log of the ith vapour pressure and let r, be the inverse of the ith temperature. The Clausius-Clapeyron equation suggests that a reasonable model for the vapour pressures over a wide range of temperatures is Yi = Bo + B1x; +6. Produce a scatter plot of the transformed data. Fit the regression model to the data and plot this regression line on the same graph with the transformed data. (c) Convert the prediction equation back to the original metric and plot this curve on the same graph with the original data.
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Given the following summary statistics, determine the regression equation used to predict y from z. By 1.65 = 82.17 L 0.11 F 21.29 76.17 Round all answers to 2 decimal places. slope y-intercept W...
-
The vapour pressure of a substance at 20.0DCis 58.0 kPa and its enthalpy of vaporization is 32.7 k] mol-1. Estimate the temperature at which its vapour pressure is 66.0 kPa.
-
The vapour pressure of a liquid in the temperature range 200 K to 260 K was found to fit the expression in (p/Torr) = 18.361 - 3036.8/ (TIK). Calculate the enthalpy of vaporization of the liquid.
-
Carbon has the electron configuration 1s 2s2p. The two unpaired electrons in the n = 2 level suggests that carbon will form two bonds. We know, however, that carbon forms four bonds in most compounds...
-
Fill in the missing amounts for a-d. ACCOUNT INFORMATION TypeRevving Account Number 234 98765 90 Biling Date 16 Aug Payment Due Date 1 Sep TRANSACTIONS I DEBITS/ CREDITS (-) 6AUG Moghan's Shop...
-
Design a serial (one bit at a time) twos complementer FSM with two inputs, Start and A, and one output, Q. A binary number of arbitrary length is provided to input A, starting with the least...
-
Using the figure in the Application "A Semiconductor Integrated Circuit Isoquant," show that as the firm employs additional fixed-proportion technologies, the firm's overall isoquant approaches a...
-
At the beginning of the current season on November 1, the ledger of Winona Sports showed Cash $3,300, Inventory $4,700, and Common Stock $8,000. The following transactions occurred during November...
-
For the experiment of rolling an ordinary pair of dice, find the probability that the sum will be less than 4 or greater than 6. The probability that the sum will be less than 4 or greater than 6 is...
-
Estimate K based on the data Differential equation: dL/dt = K(1-L) using separation of variables 1. We let L (t) be the fraction of the list already committed to memory at time t . So L =0...
-
Leakage flux in an induction motor is (A) flux that leaks through the machine (B) flux that links both stator and rotor windings (C) flux that links none of the windings (D) flux that links the...
-
Government use of legal restrictions on trade in order to achieve desired political re sults. a. comity b. transnational ins titutions c. General Agreement on Tariffs a nd Trade d. World Trade O...
-
Indicate whether each of the following statements is true or false by writing T or F in the answer c olumn. A party to a contract is released from his or her obligations if a law is passed that...
-
Indicate whether each of the following statements is true or false by writing T or F in t he a nswer c olumn. A person who defames another individual by exhibiting an insulting drawing in a public...
-
Business A pplications o f L aw Consider the numerous laws that affect businesses and evaluate the regulatory climate in which they operate. Does it seem that there are too many regulations, or too...
-
Indicate whether each of the following statements is true or false by writing T or F in t he a nswer c olumn. Another, perhaps better, name for the concept of contributory negligence might be...
-
How can accountability be operationalized through participatory governance structures, such as citizen assemblies or stakeholder engagement platforms, to enhance democratic legitimacy, public trust,...
-
An 8.0 kg crate is pulled 5.0 m up a 30 incline by a rope angled 18 above the incline. The tension in the rope is 120 N, and the crates coefficient of kinetic friction on the incline is 0.25. a. How...
-
Use the Gibbs-Duhem equation to show that the partial molar volume (or any partial molar property) of a component B can be obtained if the partial molar volume (or other property) of A is known for...
-
Suppose that a molecular orbital has the form N (O.l45A + 0.844B). Find a linear combination of the orbitals A and B that is orthogonal to this combination.
-
The speed of a certain electron is 995 km S-I. If the uncertainty in its momentum is to be reduced to 0.0010 per cent, what uncertainty in its location must be tolerated?
-
Which elements an agile software team must consider to attribute effort to a product?
-
Why is the rounded Fibonacci series used to assign story points instead of the natural numbers?
-
Variants and exception handlers are alternate flows for a use case. In which situations should one or the other be used?

Study smarter with the SolutionInn App


