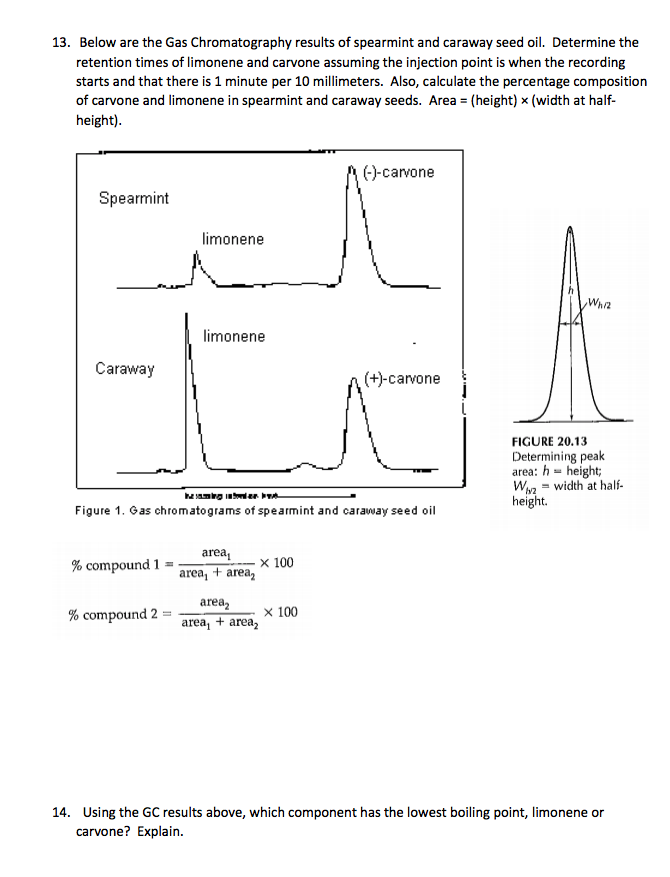
Question: 13. Below are the Gas Chromatography results of spearmint and caraway seed oil. Determine the retention times of limonene and carvone assuming the injection
13. Below are the Gas Chromatography results of spearmint and caraway seed oil. Determine the retention times of limonene and carvone assuming the injection point is when the recording starts and that there is 1 minute per 10 millimeters. Also, calculate the percentage composition of carvone and limonene in spearmint and caraway seeds. Area = (height) x (width at half- height). Spearmint Caraway % compound 1 limonene % compound 2 limonene henger kan.. Figure 1. Gas chromatograms of spearmint and caraway seed oil area area, + area area area + area x 100 (-)-carvone x 100 (+)-carvone Whiz FIGURE 20.13 Determining peak area: h= height; Wawidth at half- height. 14. Using the GC results above, which component has the lowest boiling point, limonene or carvone? Explain.
Step by Step Solution
3.41 Rating (148 Votes )
There are 3 Steps involved in it
step 1 of 2 a The relative amounts of the different substances in a GLC analysis is ... View full answer

Get step-by-step solutions from verified subject matter experts