Which of the following structures have a 1- formal charge on the sulfur atom? H-C-S: H-C-S-C-H...
Fantastic news! We've Found the answer you've been seeking!
Question:
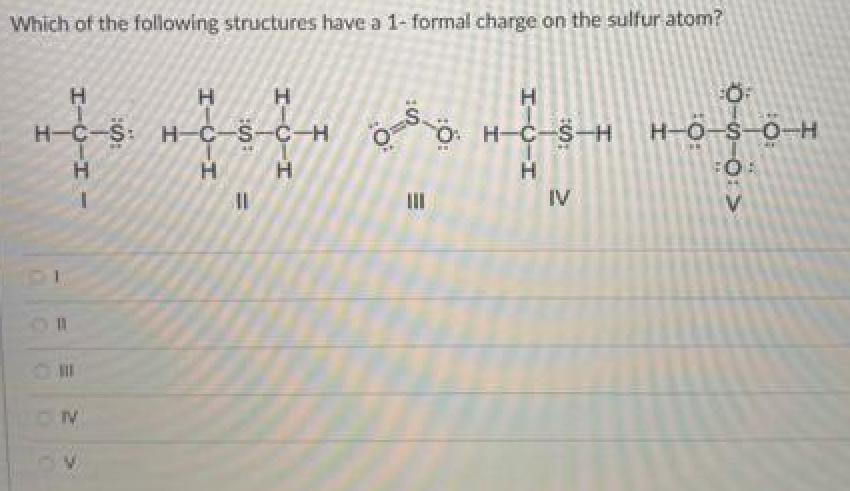
Transcribed Image Text:
Which of the following structures have a 1- formal charge on the sulfur atom? H-C-S: H-C-S-C-H S. O: H-C-S- H-O-S-O-H H. IV H. V. エーローエ エーローエ %3D エーOーエ エーUーエ ー alic Which of the following structures have a 1- formal charge on the sulfur atom? H-C-S: H-C-S-C-H S. O: H-C-S- H-O-S-O-H H. IV H. V. エーローエ エーローエ %3D エーOーエ エーUーエ ー alic
Expert Answer:
Answer rating: 100% (QA)
The formula of formal charge is Formal charge of valence electrons electrons in lone pairs 12 the nu... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Which of the following structures would allow you to know that you were upside down, eve n if you were in total darkness? a. Utricle and saccule b. Cochlea c. Semicircular canals d. Tectorial membrane
-
Which of the following structures represent mesocompounds? C (b) . (d) (c) (a) Br CH . Br .
-
Which of the following structures are identical? (Yellow-green =Cl) (a) (b) (c) (d)
-
Using the aggregate expenditures table below, answer the questions that follow. a. Compute the APC when income equals $2,300 and the APS when income equals $2,800. b. Compute the MPC and MPS. c. What...
-
Distinguish between a legal and an equitable or constructive liability. Give an example of each.
-
A rod is bent into a circular arc of radius 4 in. as shown. For the given loading, determine the internal forces at point J when 30 θ = °. 12 Ih B. 4 in.
-
You are a senior financial analyst with IBM in its capital budgeting division. IBM is considering expanding in Australia. The new facility would require an initial investment in fixed assets of 5...
-
Carmon Company is considering the addition of a new product to its cosmetics line. The company has three distinctly different options: a skin cream, a bath oil, or a hair coloring gel. Relevant...
-
1. 2. (c) Be(OH)2 (d) Sr(OH)2 Number of amphoteric compounds among the following is (a) BeO (B) Bao The stepwise formation of [Cu(NH)]* is given below: C ANH, LE [Cu(NH, ) [Cu(NH)]+NH, [Cu(NH3)2]+NH,...
-
Required - Please submit the following two documents: 1. Using Excel, submit the following: i. ii. Calculate Rashmika's minimum net income for tax purposes in accordance with the ordering provisions...
-
use the ff table for answering Tank 1 containing pure carbon monoxide (A) at 2 atm is connected to another tank, Tank 2, containing pure hydrogen gas (B) also at 72 kPa. Both tanks are at 15 o C....
-
Galina owns a home with a replacement value of $200,000. She insures it for $100,000, and has a policy that requires a minimum of 80% coverage. If her home were destroyed completely by a fire, how...
-
Suppose the annual rate of inflation in Brazil (BRL) is 11%, and the annual rate of inflation in Morocco (MAD) is 15%. If the MAD depreciates relative to the BRL by 4% in real terms, then what has...
-
Summit Inc., an Australian company, has concluded a large sale of computer systems for inventory management to a customer in Germany for 5,000,000 with payment due to be received in ninety days....
-
1. [15 points] Your boss hands you the following cash flow estimates of two mutually exclusive projects that both last for 2 years. Project A B Cost of Capital, k Investment (t=0) CF1 (t=1) CF2 (t=2)...
-
Michael is 20 years old and currently studying at university. He works part time as a waiter and saves $210 per month from his wages. Michael has so far saved $5,000 with the aim of reaching $10,000...
-
Check My Understanding Fill in the correct answers in the spaces provided. The formula A=(x+y+z)/(3) gives the average A of three values x,y, and z. Solve for x. -z -What is the value of x when the a...
-
The time to assemble the first unit on a production line is 10 hours. The learning rate is 0.94. Approximately how long will it take for the seventh unit to be assembled? The number of hours needed...
-
I.D. Chapple-Sokol, Cl Giunta, and R.G. Gordon (J Electrochem Sac 136,2993 (1989)) proposed the following radical chain mechanism for the initial stages of the gas-phase oxidation of silane by...
-
Consider the molecule CH3Cl. (a) To what point group does the molecule belong? (b) How many normal modes of vibration does the molecule have? (c) What are the symmetries of the normal modes of...
-
Show how the heat capacity of a linear rotor is related to the following sum: Where the E (J) are the rotational energy levels and g (J) their degeneracy's then go on to show graphically that the...
-
Should the whole team have decided on the team members schedule accommodations collectively? Why or why not?
-
What other solutions could you see be effective at improving employee motivation and reducing the turnover rate? Why do you believe these solutions would be useful?
-
This chapter discusses the use of job redesign to reduce turnover. Do you think this is feasible in this case? Why or why not? If so, how should the job be redesigned?

Study smarter with the SolutionInn App


