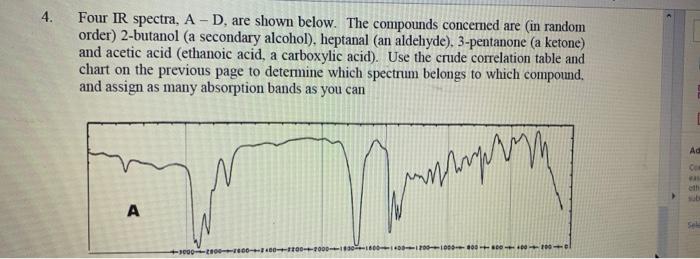
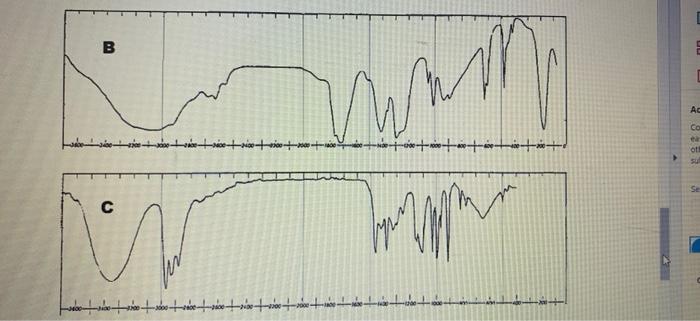
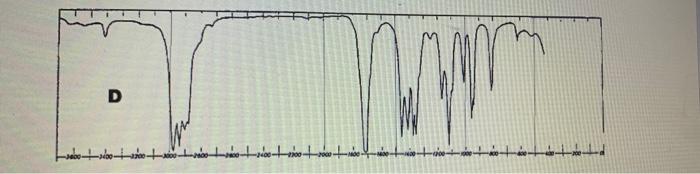
Four IR spectra, A D, are shown below. The compounds concemed are (in random order)...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Four IR spectra, A – D, are shown below. The compounds concemed are (in random order) 2-butanol (a secondary alcohol), heptanal (an aldehyde). 3-pentanone (a ketone) and acetic acid (ethanoic acid, a carboxylic acid). Use the crude correlation table and chart on the previous page to determine which spectrum belongs to which compound. and assign as many absorption bands as you can 4. Ad Co eth sub A Sele Ac Co 十章十章十年十 Se D Four IR spectra, A – D, are shown below. The compounds concemed are (in random order) 2-butanol (a secondary alcohol), heptanal (an aldehyde). 3-pentanone (a ketone) and acetic acid (ethanoic acid, a carboxylic acid). Use the crude correlation table and chart on the previous page to determine which spectrum belongs to which compound. and assign as many absorption bands as you can 4. Ad Co eth sub A Sele Ac Co 十章十章十年十 Se D
Expert Answer:
Answer rating: 100% (QA)
The IR spectrum A is the compound heptanal The structure of the heptanal is In the IR spectrum the p... View the full answer

Related Book For 

Organic Chemistry
ISBN: 978-1118133576
11th edition
Authors: Graham Solomons, Craig Fryhle, Scott Snyder
Posted Date:
Students also viewed these chemistry questions
-
The IR spectra shown next may include a carboxylic acid, an ester, an amide, a nitrile, an acid chloride, or an acid anhydride. Determine the functional group suggested by each spectrum, and list the...
-
The IR and mass spectra for three different compounds are shown in Figures 13.45-13.47. Identify each compound. a. b. c. 25 26 2.7 28 19 2000 100 43 58 100 71 85 0 20 40 60 80 100 120 m/z 13 14 15 6...
-
An infrared absorption spectrum of an organic compound is shown in the following figure. Use the characteristic group frequencies listed in Section 19.5 to decide whether this compound is more likely...
-
A gaseous mixture consists of 80.0 mole percent N2 and 20.0 mole percent O2 (the approximate composition of air). Suppose water is saturated with the gas mixture at 25C and 1.00 atm total pressure,...
-
1. a. Did the court of appeals find that Park West Bank had violated the ECOA? Explain. b. If at trial, the facts reveal that the bank employee thought Rosa was gay and demanded that he change...
-
Solve Problem 4.109 assuming that the small block C is moved and placed under edge DE at a point 0.5 ft from corner E.
-
Last year, Token Corporation reported \($1,230,000\) in sales with a cost of goods sold of \($520,000,\) and operating expenses of \($440,000.\) The increase in the firms retained earnings was...
-
Bloom Orchards produced a good crop of peaches this year. After preparing the following income statement, the company believes it should have given its No. 3 peaches to charity and saved its efforts....
-
A student addresses a bully mocking another student, " what you're doing isn't even clever or funny, everyone has a right ti express who they are.. you know like we say every morning.. liberty and...
-
For this assignment, we are going to change our point of view. You are now working for Woolly, and you are preparing Woolly's response to Mammoth's RFP to support the transition of the 12 staff from...
-
ABC Company presented current liabilities at December 31, 2X19 statement of financial position as follows: Current Liabilities Current portion of long-term debt Short-term borrowings Other financial...
-
Calculating the APR when the add-on method is used Add-on interest rates and APRs are not equivalent. This is because the add-on calculation (using the equation I = PRT , where I is the add-on...
-
4. Determine the average function and the marginal function for each of the following functions Totals: a) Total income TR = 100 Q - Q 2 b) Total cost TC = 1000+ 10 Q + .01 Q 2 c) Total profit TP =...
-
Gama Ltd. has to select a capital project between Project A and Project B. The details of both the projects are given below: Cash Flows Year Project A Project B O -2000 -2000 1 1500 150 2 700 850 3...
-
A pressurized container produced from cast iron is closed and sealed at the top with a circular plate cover made of steel. A total of 16 low-carbon martensitic Q&T steel bolts are used to join the...
-
In this assessment task students are required to develop a digital media strategy to provide information and promote a community service organisation to clients which could include: registering a...
-
Describe the three capabilities of database management systems: data definition, data dictionary, and data manipulation language. Discuss the importance of creating and using a data dictionary with a...
-
A survey of 70 college freshmen asked whether students planned to take biology, chemistry, or physics during their first year. Use the diagram to answer each question. How many of the surveyed...
-
Carry out an analysis like that shown in Fig. 9.26 and show how many peaks the signal from (b) would be split into if Jab = 2Jbc and if Jab = Jbc. (Hint: In both cases peaks will fall on top of peaks...
-
Propose structures for D, E, and F. C n peakoer0 D UV:max 230 nm H2. Pt IR: 3300 cm1, sharp UV: max 230 nm IR: -3300 cm, sharp UV: max 200 nm
-
Consider the chlorination of (S)-2-chloropentane at C4. (a) Write structural formulas for the products, showing three dimensions at all chirality centers. Give each its proper (R, S) designation. (b)...
-
Donna Corporation manufactures custom cabinets for kitchens. It uses a normalcosting system with two direct-cost categoriesdirect materials and direct manufacturing laborand one indirect-cost pool,...
-
Donna Corporation manufactures custom cabinets for kitchens. It uses a normalcosting system with two direct-cost categoriesdirect materials and direct manufacturing laborand one indirect-cost pool,...
-
Donna Corporation manufactures custom cabinets for kitchens. It uses a normalcosting system with two direct-cost categoriesdirect materials and direct manufacturing laborand one indirect-cost pool,...

Study smarter with the SolutionInn App


