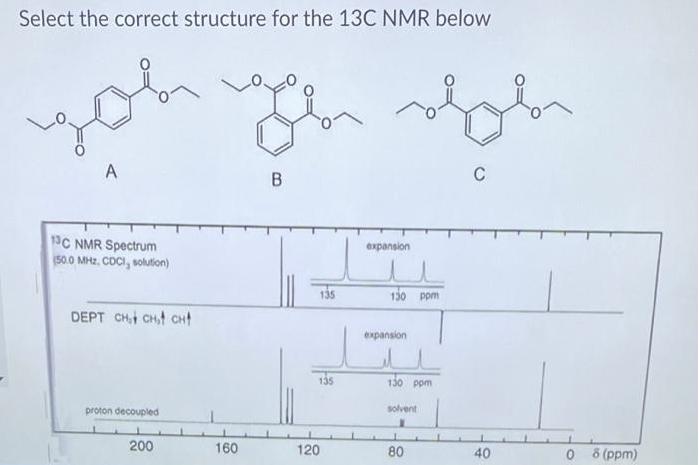
Question: Select the correct structure for the 13C NMR below you A C B 1C NMR Spectrum (50.0 MHz. CDCI, solution) expansion 135 130 ppm
Select the correct structure for the 13C NMR below you A C B 1C NMR Spectrum (50.0 MHz. CDCI, solution) expansion 135 130 ppm DEPT CHt CHt CHt expansion 135 130 pom proton decoupled solvent 200 160 120 0 8 (ppm) 80 40
Step by Step Solution
3.48 Rating (148 Votes )
There are 3 Steps involved in it
Your answer ... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (2 attachments)
63628789aaf74_235906.pdf
180 KBs PDF File
63628789aaf74_235906.docx
120 KBs Word File