Question: To analyze this data you should first identify which peak corresponds to which compound. Then calculate the percentage of each compound present for each reaction.
To analyze this data you should first identify which peak corresponds to which compound. Then calculate the percentage of each compound present for each reaction.
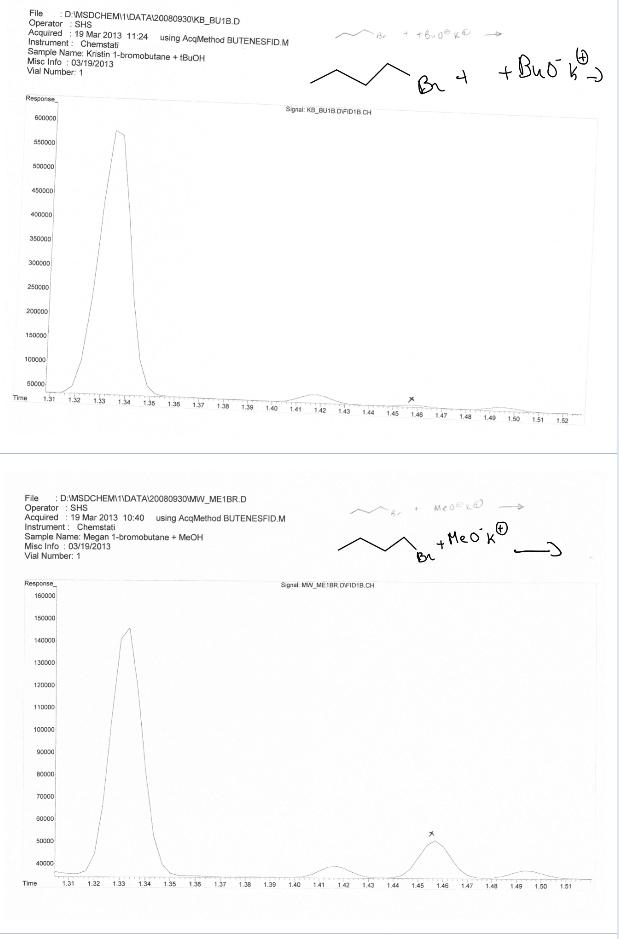
File DMSDCHEMIIDATAI20080930KB_BU1B.D SHS Operator Acquired : 19 Mar 2013 11:24 using AcgMethod BUTENESFID M Instrument: Chemstati Sampie Name: Kristin 1-bromobutane + BUOH Misc Info : 03/19/2013 Vial Number 1 + Buo Br 4 Resporse Signal: KA_BU18 DAIDIB CH 500000 4500col 400000 35000 300000 250c00 200000 150000 100000 Time 1.31 1.32 1.33 1.34 135 136 137 138 1.39 140 1.41 142 143 144 1.45 146 147 1.48 1.40 1.90 1.51 1.52 :DIMSDCHEMI1MDATA20080930MW_ME1BR.D File Operator : SHS Acquired : 19 Mar 2013 10:40 using AcqMethod BUTENESFID.M Instrument: Chemstati Sample Name: Megan 1-bromobutane + MEOH Misc Info : 03/19/2013 Vial Number: 1 Meo + Me o'kO Br Response Sgnal MW MEBR OVIDIB.CH 180000 150000 140000 130000 120000 100000 90000 70000 60000 40000 Time 1.31 1.22 1.33 1.34 135 135 137 138 1.39 1.40 1,43 141 142 1.44 1.45 1.46 147 1.4a 149 1.60 1.51
Step by Step Solution
3.42 Rating (155 Votes )
There are 3 Steps involved in it
A aitotorr U t pr swaln By t Bu 0 k 860 e majen miner Arca of major hx W12 ... View full answer

Get step-by-step solutions from verified subject matter experts


