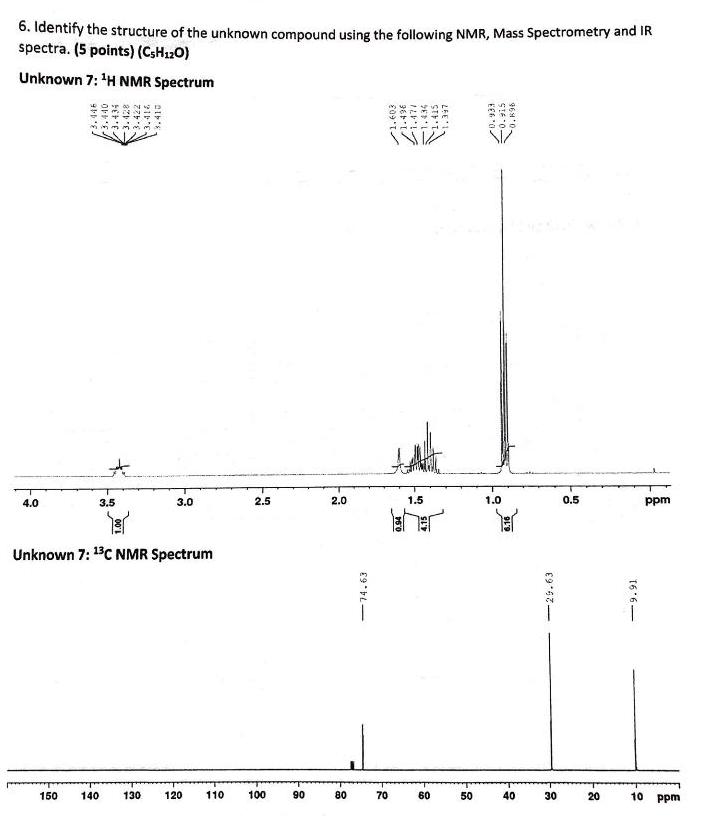
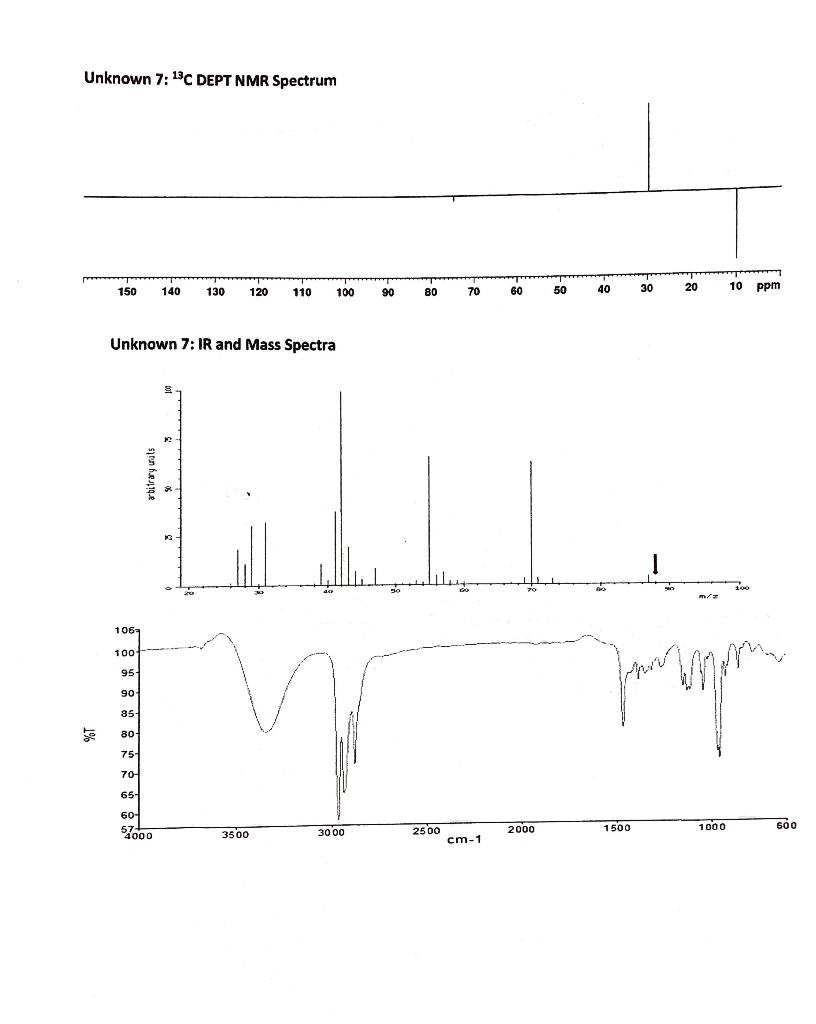
6. Identify the structure of the unknown compound using the following NMR, Mass Spectrometry and IR...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
6. Identify the structure of the unknown compound using the following NMR, Mass Spectrometry and IR spectra. (5 points) (CSH120) Unknown 7: 'H NMR Spectrum 4.0 3.5 3.0 2.5 2.0 1.5 1.0 0.5 ppm Unknown 7: 13C NMR Spectrum 150 140 130 120 110 100 06 80 70 60 50 40 30 20 10 ppm 3.440 3.434 -74.63 /1.436 1.434 1.415 LEE'1- SIE 0 -29.63 16 6- Unknown 7: 13C DEPT NMR Spectrum 50 40 30 20 10 ppm 150 140 130 120 110 100 90 80 70 60 Unknown 7: IR and Mass Spectra Foo m/a 106 100 95 90 35- 80- 75- 70 65 60- l000 25 00 2000 1500 1000 600 3500 30 00 cm-1 abitrary uats 6. Identify the structure of the unknown compound using the following NMR, Mass Spectrometry and IR spectra. (5 points) (CSH120) Unknown 7: 'H NMR Spectrum 4.0 3.5 3.0 2.5 2.0 1.5 1.0 0.5 ppm Unknown 7: 13C NMR Spectrum 150 140 130 120 110 100 06 80 70 60 50 40 30 20 10 ppm 3.440 3.434 -74.63 /1.436 1.434 1.415 LEE'1- SIE 0 -29.63 16 6- Unknown 7: 13C DEPT NMR Spectrum 50 40 30 20 10 ppm 150 140 130 120 110 100 90 80 70 60 Unknown 7: IR and Mass Spectra Foo m/a 106 100 95 90 35- 80- 75- 70 65 60- l000 25 00 2000 1500 1000 600 3500 30 00 cm-1 abitrary uats
Expert Answer:

Related Book For 

Organic Chemistry
ISBN: 978-1118133576
11th edition
Authors: Graham Solomons, Craig Fryhle, Scott Snyder
Posted Date:
Students also viewed these chemistry questions
-
Identify the structure of a compound with molecular formula C 5 H 12 that exhibits only one kind of proton. That is, all 12 protons are chemically equivalent.
-
Identify the structure of a compound with molecular formula C 9 H 20 that exhibits four CH 2 groups, all of which are chemically equivalent. How many total signals would you expect in the 1 H NMR...
-
Identify the structure of a compound with molecular formula C 9 H 10 O 2 that exhibits the following spectral data. IR: 3005 cm -1 , 1676 cm -1 , 1603 cm -1 1 H NMR: 2.6 ppm (singlet, I = 3H), 3.9...
-
Write a method leve1Order() that prints BST keys in level order: first print the root; then the nodes one level below the root, left to right; then the nodes two levels below the root (left to...
-
Define the terms covered interest arbitrage and uncovered interest arbitrage. What is the difference between these two transactions?
-
Assume that Russ Brothers did indeed fail, and that it had the following balance sheet when it was liquidated (in millions of dollars): Athe debentures are subordinated to the notes payable. The...
-
Suppose you raise this book (inertia \(m=3.4 \mathrm{~kg}\) ) from the floor to your desk, \(1.0 \mathrm{~m}\) above the floor. (a) Does the gravitational potential energy of the Earth-book system...
-
Assume the source selection criteria for evaluating proposals is as follows: Management approach, 15% Technical approach, 15% Past performance, 20% Price, 20% Interview results and samples, 30% Using...
-
Who were some of the principal Portuguese explorers? What did they achieve and what were the ramifications of these voyages for Portugal, and the world?
-
If we accept the Sharpe model as a description of expected returns, using the data in Table 16.1, find the expected return on a stock in the construction industry with the following characteristics....
-
Assume a two-period world, perfect certainty, and perfect capital market. A firm has an initial endowment of $81 million. The firm has identified the following available investment opportunities:...
-
d2x K dt m - 1 (d+ x) - 1 (d- x) (a) Classify this ODE and explain why there is little hope of solving it as is. (b) In order to solve, let's assume x < d and then replace the right-hand side...
-
Two identical loudspeakers are driven in phase by a common oscillator at 880 Hz and face each other at a distance of 1.22 m. Locate the points along the line joining the two speakers where relative...
-
Tina is aware that thousands of children starve to death every day, but only after reading a story about a single starving child did she decide to donate money to a charitable organization. Tina's...
-
What is formal differentiation and binomial theorem and how and when to use these as proof? And can you explain how to prove n,i=0(") i=n2"-1
-
1. Create a dictionary that stores item number, product name, and pricing information for a product of your choosing. Write code that will take the current price, increase it by 30% and store the...
-
A rental company spent $2500 on a new air compressor 7 years ago. The annual rental income from the compressor has been $750. The $100 spent on maintenance the first year has in- creased each year by...
-
14. In testing the existence assertion, an auditor ordinarily works from the a. Financial statements to the accounting records. b. General journal to the general ledger. c. Supporting evidence to the...
-
Myrcene, a fragrant compound found in bayberry wax, has the formula C10H16 and is known not to contain any triple bonds. (a) What is the index of hydrogen deficiency of myrcene? When treated with...
-
(a) Which of the following halides would you expect to be most reactive in an SN2 reaction? (b) In an SN1 reaction? Explain your answers. Br
-
When acetone is dissolved in water containing 18O instead of ordinary 16O (i.e., H2 18O instead of H2 16O), the acetone soon begins to acquire 18O and becomes The formation of this oxygen-labeled...
-
With regard to biological evolution, which of the following statements is incorrect? Explain why. A. During its lifetime, an animal evolves to become better adapted to its environment. B. The process...
-
Marfan syndrome is a rare inherited human disorder characterized by unusually long limbs and digits plus defects in the heart (especially the aorta) and the eyes, among other symptoms. Following is a...
-
With regard to genotypes, what is a true-breeding organism?

Study smarter with the SolutionInn App


