Choose the correct hybrid orbital diagram for TeF, which correctly shows valence electrons in this molecule....
Fantastic news! We've Found the answer you've been seeking!
Question:
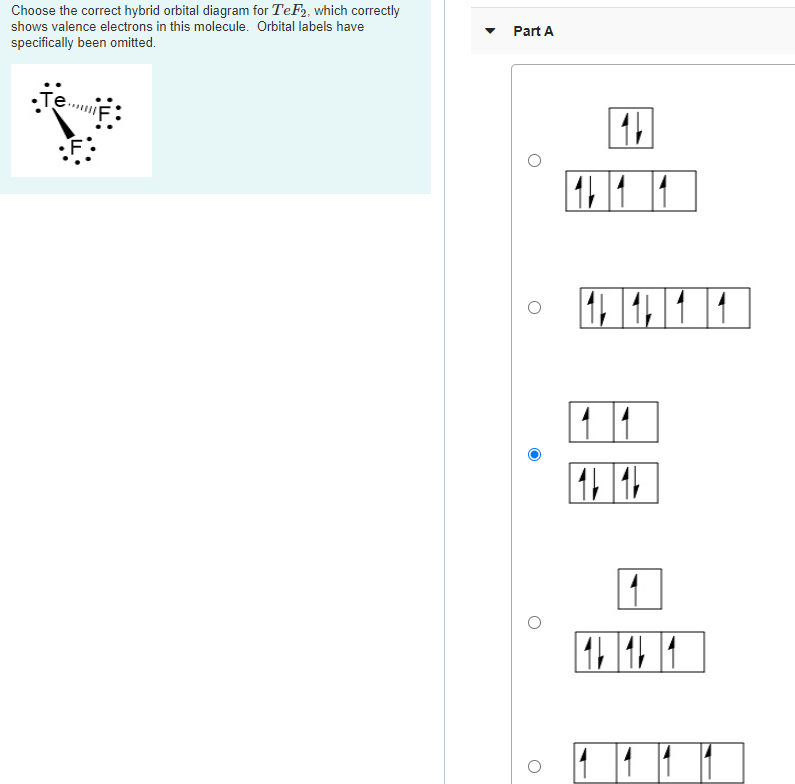
Transcribed Image Text:
Choose the correct hybrid orbital diagram for TeF₂, which correctly shows valence electrons in this molecule. Orbital labels have specifically been omitted. .Te... Part A 17 |1 1 1 1 1 1 11 161 Choose the correct hybrid orbital diagram for TeF₂, which correctly shows valence electrons in this molecule. Orbital labels have specifically been omitted. .Te... Part A 17 |1 1 1 1 1 1 11 161
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The orbital diagram that follows shows the valence electrons for a 2+ ion of an element. (a) What is the element? (b) What is the electron configuration of an atom of this element? 4d
-
The drawing below shows part of the orbital diagram for an element. (a) As drawn, the drawing is incorrect. Why? (b) How would you correct the drawing without changing the number of electrons? (c) To...
-
The following diagram shows the energies of valence molecular orbitals of boron trifluoride. The energies of three occupied orbitals (the a 2 HOMO and doubly degenerate e orbitals) are shown. The...
-
(a) lim f(x) X-C+ 5. y 7. Y 9. 2 IT - Limits and Continuity In Exercises 5-10, use the graph to determine each limit, and discuss the continuity of the function. -3+ (4,3), + + 1/2 3 4 (3, 1) (b) lim...
-
Given the following graph of a linear programming model with a single constraint and the objective function maximize Z = 30x1 + 50x2 , determine the optimal solution point: Determine the values by...
-
Jen Miller made $1,090 this week. Only social security (fully taxable) and federal income taxes attach to her pay. Miller contributes $125 each week to her companys 401(k) plan and has $40 put into...
-
Crush Autosmashers can purchase a new electromagnet for moving cars at a cost of \($20,000.\) At the end of its useful life, the electromagnet will be worth \($1,000.\) If Crushs MARR is 12...
-
(EPS with Complex Capital Structure) Amy Dyken, controller at Fitzgerald Pharmaceutical Industries, a public company, is currently preparing the calculation for basic and diluted earnings per share...
-
Assume the numbers in the diagram correspond to an area, A, in the bounded region in which they are contained. 1 9 8 2 4 5 3 What numbers make up the intersection of the circle, triangle, and square?
-
Fitz Products Inc. reported $1,075,049 profit in 2023 and declared preferred dividends of $75,100. The following changes in common shares outstanding occurred during the year. Jan. 1 78,000 common...
-
The section has tension and compression steel reinforcement. When the neutral axis depth is at infinity, c = , the section is in uniform compression, all points on the section have 0.003 compressive...
-
11. Write a Boolean equation for Z( simplify expression) < m B' D E F' N
-
An automatic egg incubator is an enclosure having controlled temperature, humidity and ventilation conditions. To stop the developing embryos from sticking to the inside of the shell, the incubator...
-
The measurement of the high hurdles is 0.65 of Robert's height in inches. Robert is 70.2 inches. How tall are the high hurdles? Round your answer to the nearest inch.
-
If there is a decrease in interest rates, what will happen for households that are net savers in the economy? They will decrease current consumption if the substitution effect is greater than the...
-
Calculate time value of money figures and use the results to support your explanations of the present and future value of your selected company. Complete your calculations on the designated tab in...
-
What is the Annual pre-tax cash flow and Minimum pre-tax return if : Project cost = $1000 Financed by: $600 debt at 9% interest (pre-tax) $400 equity with a 15% return requirement
-
Global.asax is used for: a. declare application variables O b. all other answers are wrong O c. declare global variables O d. handle application events
-
Use the LE model to describe the bonding in H2CO and C2H2. Carbon is the central atom in H2CO, and C2H2 exists as HCCH.
-
Radioactive copper-64 decays with a half-life of 12.8 days. a. What is the value of k in s-1? b. A sample contains 28.0 mg 64Cu. How many decay events will be produced in the first second? Assume...
-
What mass of barium is produced when molten BaCl2 is electrolyzed by a current of 2.50 105 A for 6.00 h?
-
In the January 1, 20X8, consolidated balance sheet, the amount of goodwill reported should be a. $0. b. $76,000. c. $95,000. d. $156,000. On January 1, 20X8, Ritt Corporation acquired 80 percent of...
-
In the December 31, 20X8, consolidated balance sheet, the amount of noncontrolling interest reported should be a. $200,000. b. $239,000. c. $251,000. d. $252,000. On January 1, 20X8, Ritt Corporation...
-
On January 1, 20X5, Post Company acquired an 80 percent investment in Stake Company. The acquisition cost was equal to Posts equity in Stakes net assets at that date. On January 1, 20X5, Post and...

Study smarter with the SolutionInn App



