Complete each of the following concentration terms with the correct ratio of quantities. mass (kg) solvent...
Fantastic news! We've Found the answer you've been seeking!
Question:
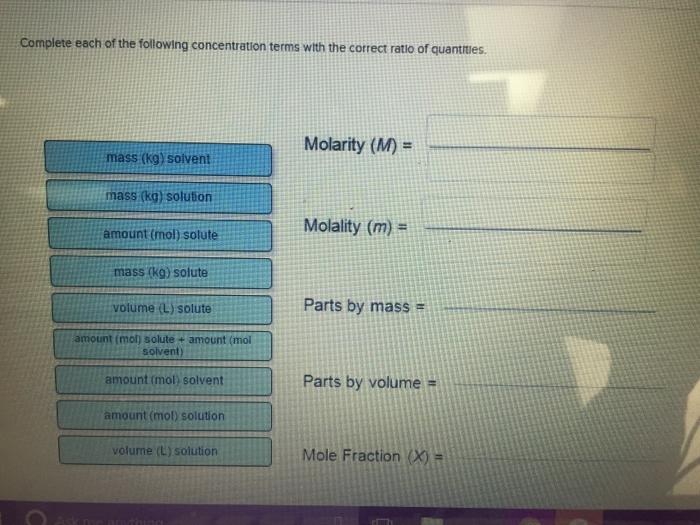
Transcribed Image Text:
Complete each of the following concentration terms with the correct ratio of quantities. mass (kg) solvent mass (kg) solution amount (mol) solute mass (kg) solute volume (L) solute amount (mol) solute + amount (mol solvent) amount (mol) solvent amount (mol) solution volume (L) solution Thing Molarity (M) = Molality (m) = Parts by mass= Parts by volume = Mole Fraction (X) = Complete each of the following concentration terms with the correct ratio of quantities. mass (kg) solvent mass (kg) solution amount (mol) solute mass (kg) solute volume (L) solute amount (mol) solute + amount (mol solvent) amount (mol) solvent amount (mol) solution volume (L) solution Thing Molarity (M) = Molality (m) = Parts by mass= Parts by volume = Mole Fraction (X) =
Expert Answer:
Answer rating: 100% (QA)
Malarity m amount mol solute vol L solution Makelin... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 9780321910417
13th Edition
Authors: Theodore E. Brown, H. Eugene LeMay, Bruce E. Bursten, Catherine Murphy, Patrick Woodward, Matthew E. Stoltzfus
Posted Date:
Students also viewed these chemistry questions
-
Complete each of the following equations: b. (CH),CHOH+ NaH a. CH,CH(OH)CH2CH3 K C. Cl e. CH3CH CHCH2SH +NaOH OH + NaOH- + NaOH
-
Complete each of the following equations: a. Butanal + excess methanol, H+ b. CH3CH(OCH3)2 + H2O, H+ c. d. + H2O, H+- O OCH + excess CH,CH,OH, H
-
Complete each of the following equations to show the conjugate acid and the conjugate base formed by proton transfer between the indicated species. Use curved arrows to show the flow of electrons,...
-
Addison, Inc. makes a single product, an indoor fireplace. Data for last year is as follows: Selling price per fireplace Manufacturing costs: Variable per unit produced: Direct materials Direct labor...
-
Star Coach, L.L.C., is in the business of converting sport utility vehicles and pickup trucks into custom vehicles. Star Coach performs the labor involved in installing parts supplied by other...
-
The following profit payoff table was presented in Problem 1. Suppose that the decision maker obtained the probability assessments P(s1) = 0.65, P(s2) = 0.15, and P(s3) = 0.20. Use the expected value...
-
Many researchers are interested in the transcription of protein-encoding genes in eukaryotes. Such researchers want to study mRNA. One method that is used to isolate mRNA is column chromatography....
-
Given the following network, determine the first activity to be crashed by the following priority rules: (a) Shortest task first (b) Minimum slack first (c) Most critical followers (d) Mostsuccessors...
-
Consider an object falling through air, where po is the density of the object, Pair is the density of the air, m is the mass of the object, A is the effective cross-sectional area of the object, or...
-
Ralph and Kathy Gump are married with one 20-year-old dependent child. Ralph earns a total of $98,000 and estimates their itemized deductions to be $28,500 for the year. Kathy is not employed. Use...
-
pleasewrite the full answer in computer typing 4. Given the process model is of the form y(k)= If the input and output data is given as k 1 2 3 2 5 6 18 C_AMO_TEM_035_02 i. ii. u(k) y(k) bz- 1 + az=...
-
List the steps necessary to avoid negative consequences of using NDT methods? List 5 negative consequences that may result from safety violation? Imagine you are construction supervisor, and you...
-
Which -(VxyP(x, y))? Explain. Ex-(VyP(x, y)) Vx-(ByP(x, y)) is logically equivalent to
-
Createe a PHP script to generate multiplication table. The user will enter the number of rows and columns (min of 1 max of 10 only) in a text box. If the user enters greater than 10 or less 2. Create...
-
The income statement of Paxson Company is presented here Paxson Company Income Statement For the Year Ended November 30,2014 Sales Revenue $7,600,000 Cost of goods sold Beginning Inventory $1,900,000...
-
Amitabh Bachan (AB), the Managing Director (MD) of Clothes Indonesia, was on a flight from Bangalore to Singapore on his way to Jakarta after a vacation. As he settled into the flight, his thoughts...
-
The owners of the store will obtain a loan for 8% for the full amount of the system. The loan would be amortized over the 5-year life of the system with payments made at the end of each year. The...
-
Selected condensed data taken from a recent statement of financial position of Morino Ltd. are as follows. MORINO LTD. Statement of Financial Position (partial) Other current assets...
-
Consider the ionic compounds KF, NaCl, NaBr, and LiCl. (a) Use ionic radii (Figure 7.8) to estimate the cationanion distance for each compound. (b) Based on your answer to part (a), arrange these...
-
Which of the following statements are true and which are false? (a) For the reaction 2A(g) + B(g) A 2 B(g) Kc and K p are numerically the same. (b) It is possible to distinguish Kc from K p by...
-
Consider the following XF 4 ions: PF 4 - , BrF 4 - , ClF 4 + , and AlF 4 - . (a) Which of the ions have more than an octet of electrons around the central atom? (b) For which of the ions will the...
-
Go to http://management.about.com/lr/project_time_ management/174690/1/ and click on Project Management on the left side of the screen. When you consider several of the articles on time management in...
-
Consider the following information that you have compiled regarding the steps needed to complete a project. You have identified all relevant steps and have made some determinations regarding...
-
Key in a search with the prompt projects in in which you select a country of interest (e.g., projects in Finland). Many of the projects generated by such a search are government-sponsored...

Study smarter with the SolutionInn App


