Which non-hydrogen groups are eclipsed in the highest energy conformation? compound (1s-3r)-3-chlorocyclohexane Highest Energy 2nd Lowest Energy
Fantastic news! We've Found the answer you've been seeking!
Question:
Which non-hydrogen groups are eclipsed in the highest energy conformation?
compound (1s-3r)-3-chlorocyclohexane
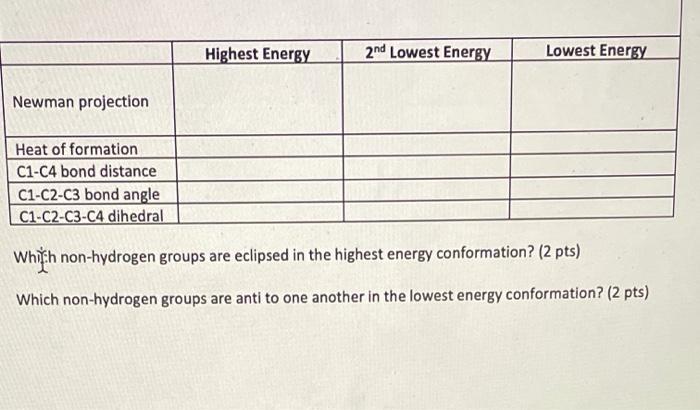
Transcribed Image Text:
Highest Energy 2nd Lowest Energy Lowest Energy Newman projection Heat of formation C1-C4 bond distance C1-C2-C3 bond angle C1-C2-C3-C4 dihedral Whith non-hydrogen groups are eclipsed in the highest energy conformation? (2 pts) Which non-hydrogen groups are anti to one another in the lowest energy conformation? (2 pts) Highest Energy 2nd Lowest Energy Lowest Energy Newman projection Heat of formation C1-C4 bond distance C1-C2-C3 bond angle C1-C2-C3-C4 dihedral Whith non-hydrogen groups are eclipsed in the highest energy conformation? (2 pts) Which non-hydrogen groups are anti to one another in the lowest energy conformation? (2 pts)
Expert Answer:
Answer rating: 100% (QA)
From the Newman Projection as illustrated below the staggered conformation of etha... View the full answer

Posted Date:
Students also viewed these chemistry questions
-
The ionization energy for a 1s electron in a silver atom is 2.462 106 kJ/ mol. a. Determine an approximate value for Zeff for the Ag 1s electron. You will first have to derive an equation that...
-
Which isotope of hydrogen has (a) The highest total binding energy and (b) The lowest average binding energy per nucleon, deuterium or tritium? Justify your answer mathematically.
-
The addition of hydrogen cyanide to benzaldehyde forms a compound called mandelonitrile. (R)-Mandelonitrile is formed from the hydrolysis of amygdalin, a compound found in the pits of peaches and...
-
The pilot of a small boat charts a course such that the boat will always be equidistant from an upcoming rock and the shoreline. Describe the path of the boat. If the rock is 2 miles offshore, write...
-
Using the formula for the security market line (Formula 177 on page 452), if the risk-free rate (RF) is 7 percent, the beta (bi) is 1.25, and the market rate of return (KM) is 11.8 percent, compute...
-
Reliable Tools, Inc., announced a 100 percent stock dividend. Determine the impact (increase, decrease, no change) of this dividend on the following: 1. Total assets 2. Total liabilities 3. Common...
-
The magnitude of the magnetic field in a magnetic resonance imaging (MRI) machine can be as great as \(B=3.0 \mathrm{~T}\). Under normal circumstances, this field cannot be shut off by just flipping...
-
Elder Corporation incurred the following transactions. 1. Purchased raw materials on account $46,300. 2. Raw Materials of $36,000 were requisitioned to the factory. An analysis of the materials...
-
Which Python data structure is constructed using the code below? a = {"a": 1, "b": 2, "c": 3}
-
Identify several ways in which you currently use accounting information in your life as a student. Also identify several situations in which, while you are still a student, you might be required to...
-
Aim of this lab is to understand and demonstrate maintenance (disassemble and assemble) of any two mechanical equipment/assemblies e.g. Head stock b. Tail stock c. Feed box d. Indexing head (lathe...
-
Prism Co, a magazine publisher, reported net profit before tax of $1,300,000 for the year ended 31 December 20x1. The only disallowed expenses were the depreciation on private motor vehicles of...
-
The Heritage Index, published yearly by the Heritage Foundation, provides a comprehensive numerical measure of overall economic freedom for countries, with specific indicators reflecting the overall...
-
The capital structure of Model Company on 31 December 20x2 is as follows: The preference shares were convertible into ordinary shares in the ratio of 1,000 preference shares for 500 ordinary shares....
-
In your opinion, is the information reported on deferred taxes relevant for decision-making? Explain.
-
Explore the reasons as to why a parent may choose not to acquire 100% of the shares in an acquiree at the date of acquisition and how it can safeguard the remaining interests by entering in...
-
In a small economy, gross investment in 2018 is $1500, consumption spending is $6000, government purchase is $1500, exports are $2000 and imports are $1000. What is nominal GDP for this economy in...
-
How has the globalization of firms affected the diversity of their employees? Why has increased diversity put an additional burden on accounting systems?
-
Consider a two-level system, with basis \(|1angle,|2angle\), and in this basis, a Hamiltonian with elements \(\left(\begin{array}{ll}1 & 1 \\ 1 & 1\end{array} ight)\). Use the first form of the...
-
Use the practical variational method for the same harmonic oscillator ground state energy, with trial wave function \(\psi_{a}(x)=e^{-a y^{2}}\).
-
Use the Ritz variational method for the harmonic oscillator, with trial wave functions \(\psi_{1}(x)=\) \(e^{-y^{2} / 2}, \psi_{2}(x)=e^{-y^{2}}, \psi_{3}(x)=e^{-2 y^{2}}\), where \(y=x \sqrt{m...

Study smarter with the SolutionInn App

