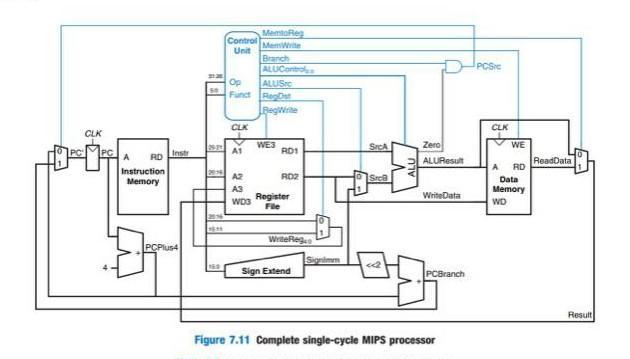
In the processor diagram Figure 7.11 on the previous page: a. Why is a mux needed...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
In the processor diagram Figure 7.11 on the previous page: a. Why is a mux needed for the A3 input to register file? b. Why does the branch control signal go to an AND gate? CLK PC A RD Instruction Memory Instr PCPlus4 *** 169 MemoReg Control MemWrite Unit Branch ALUControl Op Funct CLK A1 A2 A3 WD3 ALUSIC Regst BegWrite WE3 RD1 RDZ Register File WriteRege Sign Extend Signimm SICA Sroß ALU Zero ALUResult WriteData PCBranch Figure 7.11 Complete single-cycle MIPS processor PCS CLK WE A RD Data Memory WD ReadData Result In the processor diagram Figure 7.11 on the previous page: a. Why is a mux needed for the A3 input to register file? b. Why does the branch control signal go to an AND gate? CLK PC A RD Instruction Memory Instr PCPlus4 *** 169 MemoReg Control MemWrite Unit Branch ALUControl Op Funct CLK A1 A2 A3 WD3 ALUSIC Regst BegWrite WE3 RD1 RDZ Register File WriteRege Sign Extend Signimm SICA Sroß ALU Zero ALUResult WriteData PCBranch Figure 7.11 Complete single-cycle MIPS processor PCS CLK WE A RD Data Memory WD ReadData Result
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these computer engineering questions
-
Which of the following statements are correct about the scatter plot? 600 350 340 340 320 300 460 400 336 Othere is negative correlation between the variables. O The correlation between x and y could...
-
Complete the following ANOVA table. *Round your answer to 2 decimal places, the tolerance is +/-0.01. **Round your answer to 3 decimal places, the tolerance is +/-0.001. *** Round your answer to 2...
-
At the beginning of the year, a company's current ratio is 2.2. At the end of the year, the company has a current ratio of 2 5 Which of the following could help explain the change in the current...
-
We have titrated natural water in which the primary buffering agent is carbonate species and understood how to predict the titration curve using the equilibrium constants of those weak acids. Plot...
-
In this chapter, we wish to use a PD controller such that Gc(s) = K(s + 2). The tachometer is not used (see Figure CDP4.1). Plot the Bode diagram for the system when K = 40. Determine the step...
-
Castleman Holdings, Inc. had the following investment portfolio at January 1, 2015. During 2015, the following transactions took place. 1. On March 1, Rogers Company paid a 2 per share dividend. 2....
-
Refer to Googles financial statements in Appendix A to compute its equity ratio as of December 31, 2015, and December 31, 2014. Data From Google Financial Statement Appendix A Google Inc....
-
The comparative balance sheet of Charles Inc. for December 31, 2014 and 2013, is as follows: Additional data obtained from an examination of the accounts in the ledger for 2014 are as follows: a. The...
-
You ask your sister to look up the stock prices of two different companies: Carnival Cruise Lines (CCL) and, the world's largest toilet paper producer, Procter & Gamble (PG). How have the stock...
-
Neighborhood Supermarkets is preparing to go public, and you are asked to assist the firm by preparing its statement of cash flows for 20X1. Neighborhoods balance sheets at December 31, 20X0, and...
-
If the speed of gravity waves is the same a for light (3.00 108 m/s), as is thought to be the case, find the fundamental frequency of waves that would be detected by LIGO (distance between the two...
-
What is the straight-line method of depreciation, and how is it different from other depreciation methods? 2. What is the formula used to calculate depreciation expense under the straight-line...
-
A business retains 79% of its customers from one year to the next. If the business uses a 11% discount rate, calculate the margin multiple. Rounding: 2 decimal places.
-
All types of projects operate on a budget. Project managers need to see what the overall cost of the project is going to be from the start to the scheduled launch date. Cost Baseline is a way for PMs...
-
How do postcolonial critiques of globalization challenge dominant narratives of progress and development, foregrounding the legacies of colonialism, imperialism, and uneven power relations in shaping...
-
ACCT 326 Accounting System Review This project requires you to evaluate a PC-based accounting system to see how the system handles recording transactions, collecting data, and generating reports and...
-
Write a JavaScript function (name it: capitalizeName) to implement the following task: ** function capitalizeName(name) takes one string parameter. ** the parameter may be a full name, separated by...
-
Refer to the table to answer the following questions. Year Nominal GDP (in billions) Total Federal Spending (in billions) Real GDP (in billions) Real Federal Spending (in billions) 2000 9,817 578...
-
On January 1, 2011, Austin Auto Company decided to accumulate a fund to build an addition to its plant. The company will deposit $320,000 in the fund at each year-end, starting on December 31, 2011....
-
On December 31, 2011, Mercury Company created a fund that will be used to pay the principal amount of a $120,000 debt due on December 31, 2014. The company will make four equal annual deposits on...
-
Why is inventory an important item to both internal (management) and external users of financial statements?
-
How many structural isomers are there for hydrocarbons that have the molecular formula C 4 H 10 ? (a) none (b) one (c) two (d) three
-
Which contains more hydrogen atomsa five-carbon saturated hydrocarbon molecule or a five-carbon unsaturated hydrocarbon molecule? (a) The unsaturated hydrocarbon has more hydrogen atoms. (b) The...
-
Explain why caprylic acid, CH 3 (CH 2 ) 6 COOH, dissolves in a 5, aqueous solution of sodium hydroxide but caprylaldehyde, CH 3 (CH 2 ) 6 CHO, does not dissolve. (a) With two oxygens, the caprylic...

Study smarter with the SolutionInn App



