Consider a high-pressure vessel at 25 bar. Inside the vessel, there is a thermostat containing 1...
Fantastic news! We've Found the answer you've been seeking!
Question:
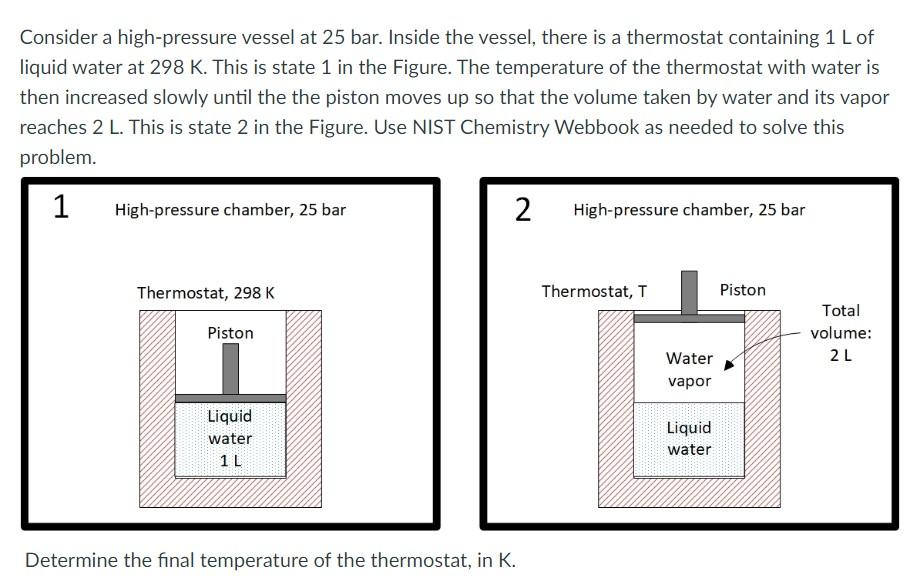
Transcribed Image Text:
Consider a high-pressure vessel at 25 bar. Inside the vessel, there is a thermostat containing 1 L of liquid water at 298 K. This is state 1 in the Figure. The temperature of the thermostat with water is then increased slowly until the the piston moves up so that the volume taken by water and its vapor reaches 2 L. This is state 2 in the Figure. Use NIST Chemistry Webbook as needed to solve this problem. 1 High-pressure chamber, 25 bar Thermostat, 298 K Piston Liquid water 1 L 2 Determine the final temperature of the thermostat, in K. High-pressure chamber, 25 bar Thermostat, T Water vapor Liquid water Piston Total volume: 2 L Consider a high-pressure vessel at 25 bar. Inside the vessel, there is a thermostat containing 1 L of liquid water at 298 K. This is state 1 in the Figure. The temperature of the thermostat with water is then increased slowly until the the piston moves up so that the volume taken by water and its vapor reaches 2 L. This is state 2 in the Figure. Use NIST Chemistry Webbook as needed to solve this problem. 1 High-pressure chamber, 25 bar Thermostat, 298 K Piston Liquid water 1 L 2 Determine the final temperature of the thermostat, in K. High-pressure chamber, 25 bar Thermostat, T Water vapor Liquid water Piston Total volume: 2 L
Expert Answer:
Answer rating: 100% (QA)
We can start by identifying the system as a closed system meaning that no matter can enter or leave ... View the full answer

Related Book For 

Elementary Principles of Chemical Processes
ISBN: 978-0471720638
3rd Edition
Authors: Richard M. Felder, Ronald W. Rousseau
Posted Date:
Students also viewed these chemical engineering questions
-
How did they determine the distance to remote galaxies? What data did they collect and what did it tell us about the expansion of the universe? What role could dark energy play in new view of the...
-
1. How strong are the competitive forces confronting J. Crew in the market for specialty retail? Do a [Michael Porter] five-forces analysis to support your answer. (see chapter 3 in the textfor...
-
Horatio Meshuggeneh has his own ideas of how to do things. For instance, when given the task of determining an oven temperature, most people would use a thermometer. Being allergic to doing anything...
-
How does Apple Inc apply Marketing and Advertising To market their product? Direct advertising Indirect Advertising Social Media (Paid Media, Owned Media, Earned Media)
-
The U.S. Census Bureau (2000 census) reported the following relative frequency distribution for travel time to work for a large sample of adults who did not work at home: a. Draw the histogram for...
-
Sam McKenzie is the founder and CEO of McKenzie Restaurants, Inc., a regional company. Sam is considering opening several new restaurants. Sally Thornton, the company's CFO, has been put in charge of...
-
Illustrate how ergonomic factors can be part of job design. LO.1
-
Ms. Mary Kelley has initial wealth W0 = $1,200 and faces an uncertain future that she partitions into two states, s = 1 and s = 2. She can invest in two securities, j and k, with initial prices of pj...
-
Costs that are considered prime costs include: a .direct materials and manufacturing overhead costs b. direct labor and direct materials costs c. direct materials only d. direct labor and...
-
During a baseball practice session, the cutoff man A executes a throw to the third baseman B. If the initial speed of the baseball is v 0 = 130 ft/sec, what launch angle is best if the ball is to...
-
Apply the classical theory of income distribution to predict the effect on the real wage and the real rental price of capital if the following events happen:(Please write down the production function...
-
CASE 7.2 Oracle Corporation: Share-Based Compensation Effects/Statement of Shareholders' Equity A sales-based ranking of software companies provided by Yahoo! Finance on November 5, Year 8, places...
-
A manufacturer of ovens sells them for $1,450 each. The variable costs are $800 per unit. The manufacturer's factory has annual fixed costs of $1,735,000. a. Given the expected sales volume of 3,100...
-
1.1 Explain the vitality of a strategy on businesses like Dell. (15) 1.2 Critically discuss the underlying objectives Dell should follow when formulating its business strategy. (20) 1.3 Discuss the...
-
Our international business plan involves exporting a sustainable apparel brand from India to UK. We will be exploring this plan in further detail below: Product/ Service: Sustainable clothing line...
-
If X is a random variable with probability density function f given by: f (x) = 4x 4x 3 when 0 x 1 and 0 otherwise, compute the following quantities: (a) The cumulative distribution function F...
-
L earning Objective 3: Describe and illustrate the financial statements of a merchandising business. a) What is the difference between a multi-step income statement and a single-step income...
-
Use the graphs of f and g to graph h(x) = (f + g) (x). To print an enlarged copy of the graph, go to MathGraphs.com. 1. 2. y 24 8. 2. -2 -2 4 6
-
In the waste liquor neutralizer, both the hydrochloric acid separated from the wetcake in the reactor centrifuge and the CO 2 generated in the resin neutralizer are neutralized with an aqueous...
-
Saturated steam at a gauge pressure of 2.0bar is to be used to heat a stream of ethane. The ethane enters a heat exchanger at 16?C and 1.5 bar gauge at a rate of 795m 3 /min and is heated at constant...
-
The indicator dilution method is a technique used to determine flow rates of fluids in channels for which devices like rotameters and orifice meters cannot be used (e.g., rivers, blood vessels, and...
-
Whales entangled in fishing gear. Refer to the Marine Mammal Science (April 2010) investigation of whales entangled by fishing gear, Exercise 10.32 (p. 552). The mean body lengths (meters) of whales...
-
Consider a completely randomized design with five treatments: A, B, C, D, and E. The ANOVA F-test revealed LO4 significant differences among the means. A multiplecomparison procedure was used to...
-
Animal-assisted therapy for heart patients. Refer to the American Heart Association Conference (Nov. 2005) study to gauge whether animal-assisted therapy can improve the physiological responses of...

Study smarter with the SolutionInn App


