Question: Consider the composition conventional feedback control system (physical process) shown in figure (1) in which a concentrated stream of control reagent containing water and
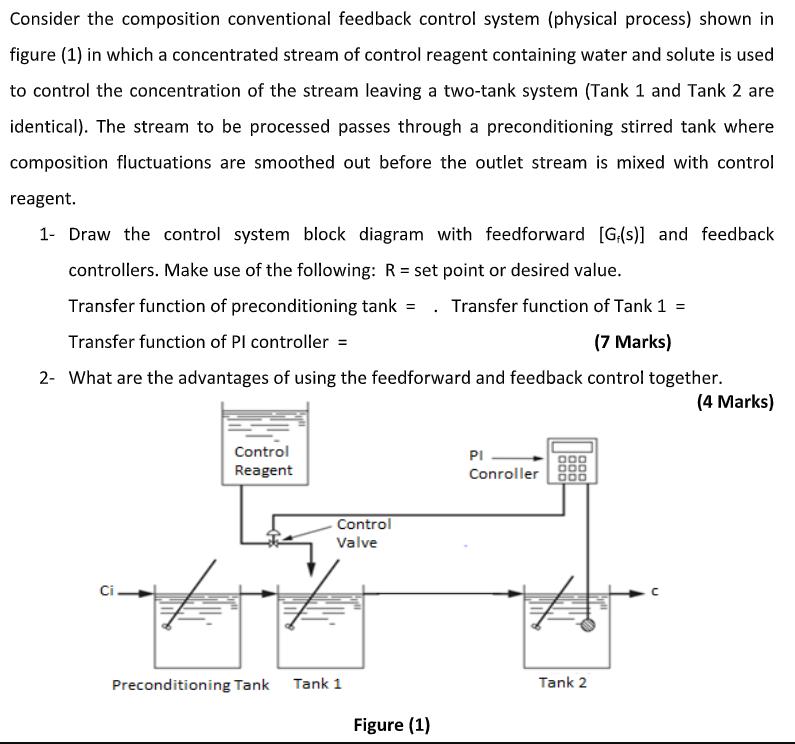
Consider the composition conventional feedback control system (physical process) shown in figure (1) in which a concentrated stream of control reagent containing water and solute is used to control the concentration of the stream leaving a two-tank system (Tank 1 and Tank 2 are identical). The stream to be processed passes through a preconditioning stirred tank where composition fluctuations are smoothed out before the outlet stream is mixed with control reagent. 1- Draw the control system block diagram with feedforward [G(s)] and feedback controllers. Make use of the following: R = set point or desired value. Transfer function of preconditioning tank = . Transfer function of Tank 1 = Transfer function of PI controller = (7 Marks) 2- What are the advantages of using the feedforward and feedback control together. (4 Marks) Control PI Reagent Conroller Control Valve Ci. Preconditioning Tank Tank 1 Tank 2 Figure (1)
Step by Step Solution
3.54 Rating (144 Votes )
There are 3 Steps involved in it
Part1 A feed forward sometimes written feedforward is an element or pathway within a control system that passes a controlling signal from a source in its external environment to a load elsewhere in it... View full answer

Get step-by-step solutions from verified subject matter experts


