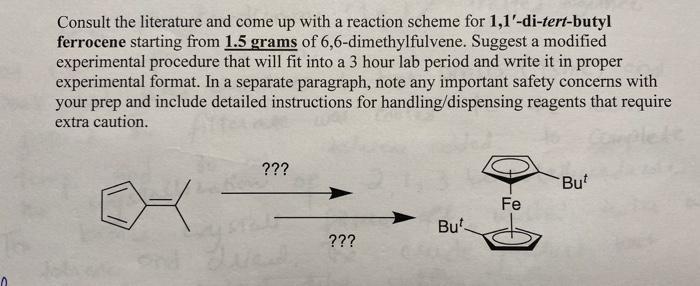
Consult the literature and come up with a reaction scheme for 1,1'-di-tert-butyl ferrocene starting from 1.5...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Consult the literature and come up with a reaction scheme for 1,1'-di-tert-butyl ferrocene starting from 1.5 grams of 6,6-dimethylfulvene. Suggest a modified experimental procedure that will fit into a 3 hour lab period and write it in proper experimental format. In a separate paragraph, note any important safety concerns with your prep and include detailed instructions for handling/dispensing reagents that require extra caution. ??? ??? But Fe But Consult the literature and come up with a reaction scheme for 1,1'-di-tert-butyl ferrocene starting from 1.5 grams of 6,6-dimethylfulvene. Suggest a modified experimental procedure that will fit into a 3 hour lab period and write it in proper experimental format. In a separate paragraph, note any important safety concerns with your prep and include detailed instructions for handling/dispensing reagents that require extra caution. ??? ??? But Fe But
Expert Answer:
Answer rating: 100% (QA)
Solution An exceptionally simple and efficient synthesis of its oxidative transformations A new ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A company has come up with a new selection test and decides to try it out on some of its current workers before giving it to job applicants. A group of its current workers volunteered to take the...
-
A pharmaceutical manufacturer has come up with a new drug intended to provide greater headache relief than the old formula. Of 250 patients treated with the previous medication, 130 reported fast...
-
EarthTones, Inc., was a wholly owned subsidiary of Cutlinger Corporation. Several years ago, EarthTones acquired numerous oil leases and began exploration activities to determine their suitability...
-
What are the disadvantages of secondary data?
-
The following transactions occurred for Molina Engineering: Requirements 1. Open the following T-accounts for Molina Engineering: Cash; Accounts Receivable; Supplies; Equipment; Accounts Payable;...
-
With reference to the preceding exercise, construct a 95% confidence interval for \(\alpha\).
-
Queensland Company reports the following operating results for the month of April. Management is considering the following course of action to increase net income: Reduce the selling price by 4%,...
-
Blossom Company is considering two different, mutually exclusive capital expenditure proposals. Project A will cost $450,000, has an expected useful life of 11 years, a salvage value of zero, and is...
-
Lucy is in charge of planning and coordinating a project. Following is the activity information for this project. Time (weeks) Activity Preceded By Optimistic Most Likely Pessimistic Lucy is in...
-
Water weighs about 8.34 pounds per gallon. About how many ounces per gallon is the weight of the water.
-
Why is it useful to integrate the collection of online and offline metrics?
-
Identify two examples of branding in financial services (e.g., specific types of retail bank accounts or insurance policies) and define their characteristics. How meaningful are these brands likely...
-
Describe four different types of site on which online display advertising for a car manufacturers site could be placed.
-
Visit the facilities of two competing service firms in the same industry (e.g., banks, restaurants, or gas stations) that you believe have different approaches to service. Compare and contrast their...
-
Compare the effectiveness of different methods of online advertising including display advertisements, paid search marketing and affiliate marketing.
-
What are the mechanisms underlying the coordination of cell cycle progression with cellular metabolism, energy production, and nutrient availability, and how do metabolic checkpoints and signaling...
-
Consider the combustion of methanol below. If 64 grams of methanol reacts with 160 grams of oxygen, what is the CHANGE in volume at STP. 2CH3OH(g) + 3O2(g) 2CO2(g) + 4H2O(1) The volume decreases by...
-
Analysis of a time series consisting of weekly visits (in thousands) to a corporate website has led to the following autoregressive forecasting equation: t = 34.50 + 0.58yt1 0.72yt2. If there were...
-
Based on the sample results in Exercise 10.109, construct and interpret the 95% confidence interval for the population proportion. Is the hypothesized proportion (0.25) within the interval? Given the...
-
An investment counselor would like to meet with 12 of his clients on Monday, but he has time for only 8 appointments. How many different combinations of the clients could be considered for inclusion...
-
In March 2015, a Nielsen global online survey "found that consumers are increasingly willing to pay more for socially responsible products." Over 30,000 people in 60 countries were polled about their...
-
Proportion of US adults who own a cell phone. In a survey of 1006 US adults in 2014, 90% said they had a cell phone. Give the correct notation for the quantity described and give its value.
-
One of the many wonderful things about studying statistics is that graduate programs in statistics often pay their graduate students, which means that many graduate students in statistics are able to...

Study smarter with the SolutionInn App


