Based on what we learned about the color of organic molecules, do the spectra of the structures
Fantastic news! We've Found the answer you've been seeking!
Question:
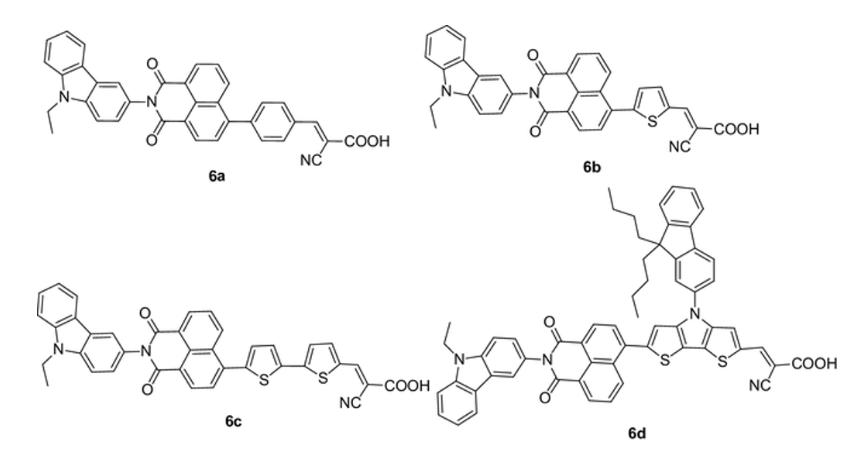
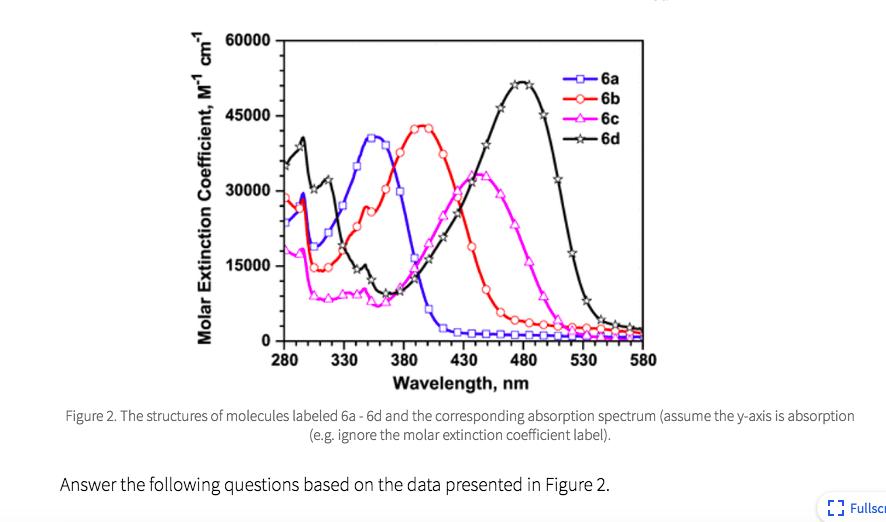
Based on what we learned about the color of organic molecules, do the spectra of the structures labeled 6a - d follow the trend you expect? Why?

Transcribed Image Text:
B C D E No. The number of bonds contributing to the largest conjugated part of the molecules, from the shortest to the longest, are 6b, 6a, 6c, and 6d. Therefore, 6b would have the shortest wavelength of maximum absorption and 6d would have the longest wavelength of maximum absorption. However, the experimental spectrum indicates that 6a has the shortest wavelength of the series. Yes. The number of bonds contributing to the largest conjugated part of the molecules, from the shortest to the longest, are 6a, 6b, 6c, and 6d. Therefore, 6a would have the shortest wavelength of maximum absorption and 6d would have the longest wavelength of maximum absorption. The experimental spectrum supports this trend. No. The intensity of the spectrum corresponds to the number of double bonds in the molecule. 6d has the highest number of double bonds and has the spectrum with the highest intensity of the four molecules. However, 6c has the second highest number of double bonds and has the lowest intensity of the four molecules. Therefore, the experimental spectra do not support the current understanding of the molecular properties of organic molecules that contribute to their color. Yes. The intensity of the spectrum corresponds to the number of double bonds in the molecule. 6d has the highest number of double bonds and has the spectrum with the highest intensity of the four molecules. 6b has the lowest number of double bonds and has the lowest intensity of the four molecules. Therefore, the experimental spectra do support the current understanding of the molecular properties of organic molecules that contribute to their color. No. The molecules do not differ in ways that we have discussed should impact the visible spectrum of organic molecules. However, the nitrogen and/or sulfur content increases from 6a to 6d and correlates with the trend in wavelength increases (i.e. more electronegative atoms increases the wavelength). Therefore, the experimental spectra compared to the molecule structure supports a new understanding of the molecular properties of organic molecules that contribute to their color. B C D E No. The number of bonds contributing to the largest conjugated part of the molecules, from the shortest to the longest, are 6b, 6a, 6c, and 6d. Therefore, 6b would have the shortest wavelength of maximum absorption and 6d would have the longest wavelength of maximum absorption. However, the experimental spectrum indicates that 6a has the shortest wavelength of the series. Yes. The number of bonds contributing to the largest conjugated part of the molecules, from the shortest to the longest, are 6a, 6b, 6c, and 6d. Therefore, 6a would have the shortest wavelength of maximum absorption and 6d would have the longest wavelength of maximum absorption. The experimental spectrum supports this trend. No. The intensity of the spectrum corresponds to the number of double bonds in the molecule. 6d has the highest number of double bonds and has the spectrum with the highest intensity of the four molecules. However, 6c has the second highest number of double bonds and has the lowest intensity of the four molecules. Therefore, the experimental spectra do not support the current understanding of the molecular properties of organic molecules that contribute to their color. Yes. The intensity of the spectrum corresponds to the number of double bonds in the molecule. 6d has the highest number of double bonds and has the spectrum with the highest intensity of the four molecules. 6b has the lowest number of double bonds and has the lowest intensity of the four molecules. Therefore, the experimental spectra do support the current understanding of the molecular properties of organic molecules that contribute to their color. No. The molecules do not differ in ways that we have discussed should impact the visible spectrum of organic molecules. However, the nitrogen and/or sulfur content increases from 6a to 6d and correlates with the trend in wavelength increases (i.e. more electronegative atoms increases the wavelength). Therefore, the experimental spectra compared to the molecule structure supports a new understanding of the molecular properties of organic molecules that contribute to their color.
Expert Answer:
Answer rating: 100% (QA)
B Yes The number of bonds contributing to the largest conjugated part of the molecules from the shortest to the longest are 6a 6b 6c and 6d Therefore 6a would have the shortest wavelength of maximum a... View the full answer

Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Posted Date:
Students also viewed these accounting questions
-
Answer the following questions based on the data from Problem 14.73. a. Calculate the slope and the y intercept for the regression equation. b. Predict the order size of a 32 year old customer. c....
-
Answer the following questions based on the data from Problem 14.73. a. Calculate the coefficient of determination. b. Test the significance of the population coefficient of determination using =...
-
Answer the following questions based on the data from Problem 14.73. a. Test for the significance of the regression slope using = 0.05. b. Construct a 95% confidence interval for the population...
-
1. Consider the following problem: Suppose that the interest rate in this year is 5% and all investors expect that interest rates for the next 3 years will be as follows: Year 1 2 3 Forward Interest...
-
Provide a mechanism for the following reaction. NaOH H20
-
The following are an income statements and balance sheets for General Foods Ltd. REQUIRED a. Use professional judgment in deciding on the preliminary judgment about materiality for profit, current...
-
What are some potential disadvantages of using a packaged application to help automate the employee goal-setting process?
-
The following transactions of Dunn Miles occurred during 2012: Apr 30 Miles is party to a patent infringement lawsuit of $230,000. Miless attorney is certain it is remote that Miles will lose this...
-
How could EMS services play a role in advancing the CCMC (Committee on the cost of medical care) guideline advocating for the extension of all basic public health services to the entire population?
-
On January 30 of the current year, Ashton Corporation purchased from Cathy 10% of Todd Corporation stock for $250,000 in cash. On May 30 of the following year, Andrea and Bill each exchange one-half...
-
The following questions are based on Air Canada's March-April 2022 Inflight Magazine: In a new browser, please copy and paste this link:...
-
Complete the table about estimation of asset acquisition. a. In your own words, describe each listed data used in estimation of asset acquisition. b. In your own words, explain how the data is used...
-
calculate the accessible income INCOME $ $ C - OTHER SALES OF GOODS AND SERVICES Sales of goods and services to Australian customers 525000 Exported sales of goods and services 65000 590000 H - TOTAL...
-
describe the corporation j McIntyre machinery and its activities or contacts with he unites states.
-
Polly's Pet products has a balance sheet and a statement of cash flow. In the balance sheet the value of cash under current assets is missing. How can I find the value for cash using the statement of...
-
Match the action to the correct corresponding step of the accounting cycle. Record transactions in journals. Analyze source documents. Take a trial balance. Analyze financial statements. Transfer...
-
It is easier to take in hand, more perilous to conduct, and more uncertain in its success than to take the lead in introducing a new order of things. Because the innovator has enemies in all those...
-
1-Stern observed all of the following results EXCEPT _______ in his experiment. A-one of the recombinant phenotypes was associated with an X chromosome of normal length B-the number of car, B+ male...
-
A 5.0-cm-diameter coil has 20 turns and a resistance of 0.50 . A magnetic field perpendicular to the coil is B = 0.020t + 0.010t 2 , where B is in tesla and t is in seconds. a. Find an expression for...
-
A cart that is rolling at constant velocity on a level table fires a ball straight up. a. When the ball comes back down, will it land in front of the launching tube, behind the launching tube, or...
-
How much energy is stored in a 3.0-cm-diameter, 12-cm long solenoid that has 200 turns of wire and carries a current of 0.80 A?
-
a. For the allowed energies of a particle in a box to be large, should the box be very big or very small? Explain. b. Which is likely to have larger values for the allowed energies: an atom in a...
-
The molecules in the rods and cones in the eye are tuned to absorb photons of particular energies. The retinal molecule, like many molecules, is a long chain. Electrons can freely move along one...
-
What was the approximate activity of the plutonium source at the start of the mission? A. \(2 \times 10^{21} \mathrm{~Bq}\) B. \(2 \times 10^{19} \mathrm{~Bq}\) C. \(2 \times 10^{17} \mathrm{~Bq}\)...

Study smarter with the SolutionInn App


