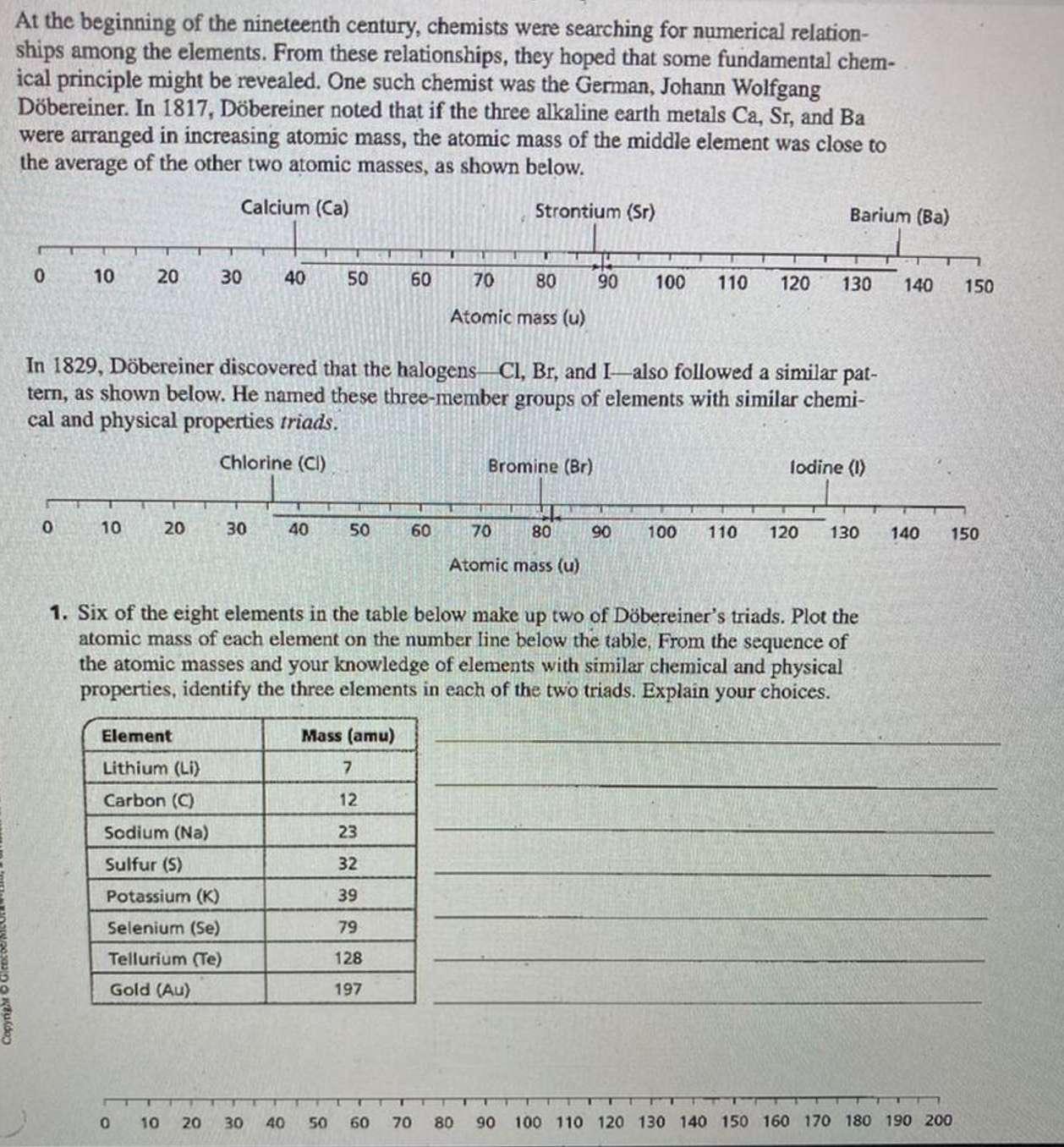
Copyright At the beginning of the nineteenth century, chemists were searching for numerical relation- ships among...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Copyright At the beginning of the nineteenth century, chemists were searching for numerical relation- ships among the elements. From these relationships, they hoped that some fundamental chem- ical principle might be revealed. One such chemist was the German, Johann Wolfgang Döbereiner. In 1817, Döbereiner noted that if the three alkaline earth metals Ca, Sr, and Ba were arranged in increasing atomic mass, the atomic mass of the middle element was close to the average of the other two atomic masses, as shown below. Calcium (Ca) Strontium (Sr) 0 10 0 20 10 20 30 Element Lithium (Li) Carbon (C) Sodium (Na) Sulfur (5) 40 30 Potassium (K) Selenium (Se) Tellurium (Te) Gold (Au) 50 In 1829, Döbereiner discovered that the halogens Cl, Br, and I also followed a similar pat- tern, as shown below. He named these three-member groups of elements with similar chemi- cal and physical properties triads. Chlorine (Cl) 40 0 10 20 30 40 50 60 Mass (amu) 7 12 23 32 39 79 128 197 60 T U 70 80 Atomic mass (u) 50 60 70 U Bromine (Br) 70 80 Atomic mass (u) 80 90 100 110 120 1. Six of the eight elements in the table below make up two of Döbereiner's triads. Plot the atomic mass of each element on the number line below the table, From the sequence of the atomic masses and your knowledge of elements with similar chemical and physical properties, identify the three elements in each of the two triads. Explain your choices. Barium (Ba) 90 130 lodine (1) 100 110 120 130 140 150 140 150 T 90 100 110 120 130 140 150 160 170 180 190 200 Copyright At the beginning of the nineteenth century, chemists were searching for numerical relation- ships among the elements. From these relationships, they hoped that some fundamental chem- ical principle might be revealed. One such chemist was the German, Johann Wolfgang Döbereiner. In 1817, Döbereiner noted that if the three alkaline earth metals Ca, Sr, and Ba were arranged in increasing atomic mass, the atomic mass of the middle element was close to the average of the other two atomic masses, as shown below. Calcium (Ca) Strontium (Sr) 0 10 0 20 10 20 30 Element Lithium (Li) Carbon (C) Sodium (Na) Sulfur (5) 40 30 Potassium (K) Selenium (Se) Tellurium (Te) Gold (Au) 50 In 1829, Döbereiner discovered that the halogens Cl, Br, and I also followed a similar pat- tern, as shown below. He named these three-member groups of elements with similar chemi- cal and physical properties triads. Chlorine (Cl) 40 0 10 20 30 40 50 60 Mass (amu) 7 12 23 32 39 79 128 197 60 T U 70 80 Atomic mass (u) 50 60 70 U Bromine (Br) 70 80 Atomic mass (u) 80 90 100 110 120 1. Six of the eight elements in the table below make up two of Döbereiner's triads. Plot the atomic mass of each element on the number line below the table, From the sequence of the atomic masses and your knowledge of elements with similar chemical and physical properties, identify the three elements in each of the two triads. Explain your choices. Barium (Ba) 90 130 lodine (1) 100 110 120 130 140 150 140 150 T 90 100 110 120 130 140 150 160 170 180 190 200
Expert Answer:
Answer rating: 100% (QA)
Dobereiners triads represent a classification system for elements that significantly contributed to ... View the full answer

Related Book For 

Posted Date:
Students also viewed these mathematics questions
-
The article below focuses on this issue. According to the article, total robotic sales for the first nine months of 2021 reached a record $1.48 billion. Labor shortages are causing companies in...
-
It's October 10, 2021, and you, CPA, work as an audit senior at Chow and Co. LLP. One of the firm's long-time clients, Tyler Wallace, owns a group of companies, and recently added a new addition to...
-
Write a literature review for your study. See below for an example of a literature review. Your literature review should provide both analysis and synthesis of previous studies as related to the...
-
Capital Inc. has prepared the operating budget for the first quarter of 2015. They forecast sales of $50,000 in January, $60,000 in February, and $70,000 in March. Variable and fixed expenses are as...
-
Let f, g, h, k: NN where f(n) = 3n, g(n) = [n/3], h(n) = [(n + 1)/3], and k(n) = (n + 2)/3], for each n N. (a) For each n N what are (g o f)(h o f) and {k o f){n?)l (b) Do the results in part (a)...
-
Problem 1 Sixty percent of adults over 60 have received a vaccine for shingles. The vaccine is not perfect, and sometimes vaccinated people get the disease. Suppose 20% of adults over 60 have had the...
-
The receiving department at Culp Electronics Company processes inventory deliveries upon arrival by means of online data terminals located on the receiving dock. Each inventory receipt entered into...
-
For U. S. public corporations, SFAS 123R required expensing of ESOs. However, the exposure draft of this standard met considerable opposition, mostly from large technology companies. These companies...
-
This is my second round posting this question. I really want to know how to do this. Please explain how to in-put answers in cell formula IE 09-02 (Static) Using Excel to Calculate and Prepare the...
-
Mary and John Parent have a 12 year old son in middle school in the Contra Costa School District. The child "has a long standing formal diagnosis of Autism Spectrum Disorder, which impairs his social...
-
Michael Sanchez purchased a condominium for $86,000. He made a 20% down payment and financed the balance with a 30 year, 5% fixed-rate mortgage. (Round your answers to the nearest cent. Use this...
-
A piezoresistive sensor has a measurement error of 1 mV. During the measurement the temperature fluctuation and its self-heating cause additional measurement errors of 0.2 mV and 0.05 mV...
-
2. You would like to have $500,000 when you retire in 15 years. How much should you invest each quarter if you can earn a rate of 6.8 % compounded quarterly? (4 pts)
-
Specifically, I want to discuss preventing damage to evidence. When handling evidence, we need to consider several factors, including, but not limited to, the authenticity of the evidence and the...
-
You are going to borrow $2000 from your parents to put towards a down payment on your first car. Your parents are going to charge you 3% simple interest. If you are going to pay them back at the end...
-
Steve is charged with speeding and reckless driving. The incident caused another car to get run off the road. Steve is given a choice to plead guilty or nolo contendere. You are his defense attorney,...
-
You are running an arbitrage-based hedge fund focusing on merger transactions. Company XYZ has just received a tender offer for $47 per share from the management of AcquisiCorp. For the past two...
-
Describe basic managerial approaches to implementing controls and how these are implemented.
-
The chief financial officer of a firm presented the CEO with a set of financial statements showing $2,234 million in after-tax operating income. This number yielded a return on beginning-of-period...
-
Xerox Corporation is a long-established company whose very name has been lent to the process of copying documents. The firm develops copying technology through an extensive research program and...
-
Comment on the following statement. The challenge in measuring the required return for investing is to measure the size of the risk premium over the risk-free rate, but the capital asset pricing...
-
Create a similar barplot as in Figure 1. 5, but now plot the corresponding proportions of males and females in each of the three situation categories. That is, the heights of the bars should sum up...
-
Import the data set EuStockMarkets from the same website as the iris data set above. The data set contains the daily closing prices of four European stock indices during the 1990s, for 260 working...
-
The iris data set, mentioned in Section1.1, contains various features, including 'Petal.Length' and 'Sepal.Length', of three species of iris: setosa, versicolor, and virginica. (a) Load the data set...

Study smarter with the SolutionInn App


