Create the flammability diagram for the benzene/nitrogen/oxygen system. Benzene has a fla point of -11.6 C,...
Fantastic news! We've Found the answer you've been seeking!
Question:
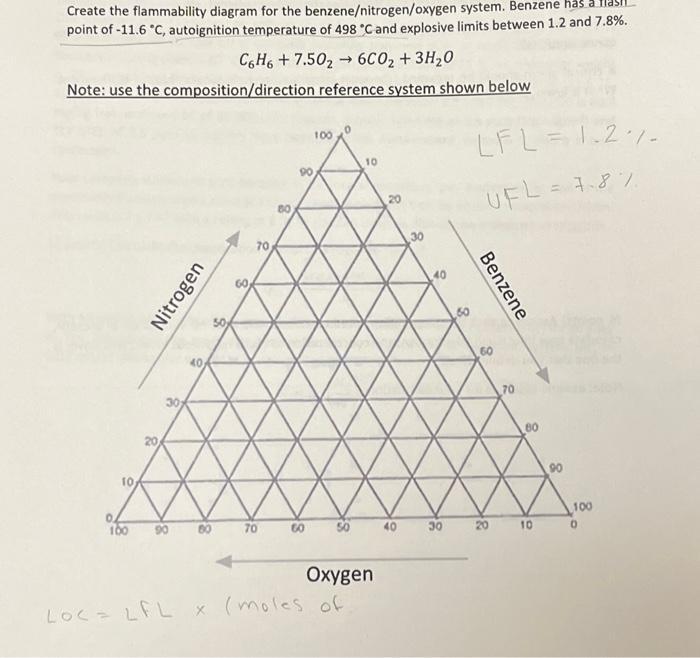
Transcribed Image Text:
Create the flammability diagram for the benzene/nitrogen/oxygen system. Benzene has a fla point of -11.6 °C, autoignition temperature of 498 "C and explosive limits between 1.2 and 7.8%. C6H6 +7.5026C0₂ + 3H₂0 Note: use the composition/direction reference system shown below 10 100 90 8 Nitrogen 80 60 70 70 80 90 60 100 10 Oxygen LOC = LFL x (moles of 20 40 30 30 60 LFL-1.2- UFL = 7.8% Benzene 60 20 70 80 10 90 100 0 Create the flammability diagram for the benzene/nitrogen/oxygen system. Benzene has a fla point of -11.6 °C, autoignition temperature of 498 "C and explosive limits between 1.2 and 7.8%. C6H6 +7.5026C0₂ + 3H₂0 Note: use the composition/direction reference system shown below 10 100 90 8 Nitrogen 80 60 70 70 80 90 60 100 10 Oxygen LOC = LFL x (moles of 20 40 30 30 60 LFL-1.2- UFL = 7.8% Benzene 60 20 70 80 10 90 100 0
Expert Answer:
Answer rating: 100% (QA)
Accumulate Fundamental Information Unsound Attributes of Benzene 1 LFL Lower Combustibility Breaking point 12 Least benzene focus for ignition 2 UFL Upper Combustibility Cutoff 78 Most extreme benzene ... View the full answer

Related Book For 

Posted Date:
Students also viewed these law questions
-
8(k) sf (k) a) Given the figure above, briefly explain why the identified capital-labor ratio cannot be a countries' steady state level. b) How can the government achieve the identified capital-labor...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
John Deer began a restaraunt consulting company. Below are events and transactions that occurred during the first month of operation. May 1 John Deer invested $38,000 cash to start a consulting...
-
What do U-235 and Pu-239 have in common?
-
Positive Accounting Theory utilises the concept of political costs. Briefly define political costs. What actions might a firms management undertake in an attempt to minimise the political costs that...
-
The suitable pump for flood control and irrigation applications is (a) reciprocating (b) centrifugal (c) axial flow (d) mixed flow
-
Valient Online Products is considering adopting the balanced scorecard and has compiled the following list of possible performance measures. Select the balanced scorecard perspective that best...
-
BMX Company has one employee. FICA Social Security taxes are 6.2% of the first $137,700 paid to its employee, and FICA Medicare taxes are 1.45% of gross pay. For BMX, its FUTA taxes are 0.6% and SUTA...
-
(a) Without referring back, make a list of the most important items you learned in this chapter. (b) Overall, what do you believe were the three major purposes of the chapter?
-
What does "impossible trinity" mean? Explain how they are important when setting a foreign exchange rate policy. ii. "An option contract which is arranged over the counter provides not only the...
-
Discuss which cost structure would be most beneficial (pure variable or pure fixed) when sales volume is increasing and when sales volume is decreasing.
-
If Frank drove 158 miles last week and this week he drove c miles. Using c how would I write an expression for the total number of hours he drove in two weeks.
-
The average annual returns (in percentages) on NASDAQ and S&P 500 for the past 10 years are given below: Year NASDAQ S&P 500 2012 15.91 16 2013 38.32 32.4 2014 13.4 13.7 2015 5.73 1.4 2016 7.5 12...
-
Find the slope of the tangent line at point (-2,2) on the curve f(x) = 2 x 2 + 3x using First Principles.
-
Consider some quadratic function. Given that the x intercepts to the quadratic function are x = 8 and x = 3 , write the equation that has these intercepts and opens down. Be sure that the leading...
-
You have been the auditor for Bob Edward Thomas Ltd. (BET) for twelve years, including the current year ended December 31, 2021. BET is a public company listed on the TSX. In the current year, an...
-
Heineken N.V., a global brewer based in the Netherlands, reports the following balance sheet accounts for the year ended December 31, 2016 (euros in millions). Prepare the balance sheet for this...
-
The volumetric flow rate through a reactor is 10 dm 3 /min. A pulse test gave the following concentration measurements at the outlet: a. Plot the external-age distribution E(t) as a function of time....
-
A diesel engine does not use spark plugs to ignite the fuel and air in the cylinders. Instead, the temperature required to ignite the fuel occurs because the pistons compress the air in the...
-
Repeat parts (a)(c) of Problem P4-11B for the reaction 3A C Pure A enters at 400 K, 10 atm and the equilibrium constant is K C = 2.5 (dm 3 /mol) 2 . Compare the equilibrium conversions in Problems...
-
In the second quarter of 2021, personal consumption expenditures, exports, and imports increased. Investment and government expenditure decreased. Real GDP increased by 6.5 percent following a 6.3...
-
When real GDP increased in the second quarter of 2021, consumption expenditure, exports, and imports increased. Fixed investment decreased, which included a decrease in business inventory investment....
-
Real exports of goods and services increased 6 percent in the second quarter of 2021, compared with a decrease of 2.9 percent in the first quarter of 2021. Real imports of goods and services...

Study smarter with the SolutionInn App


