Daily demand (average) Standard deviation of Demand = Length of replenishment cycle (average) = Standard deviation...
Fantastic news! We've Found the answer you've been seeking!
Question:
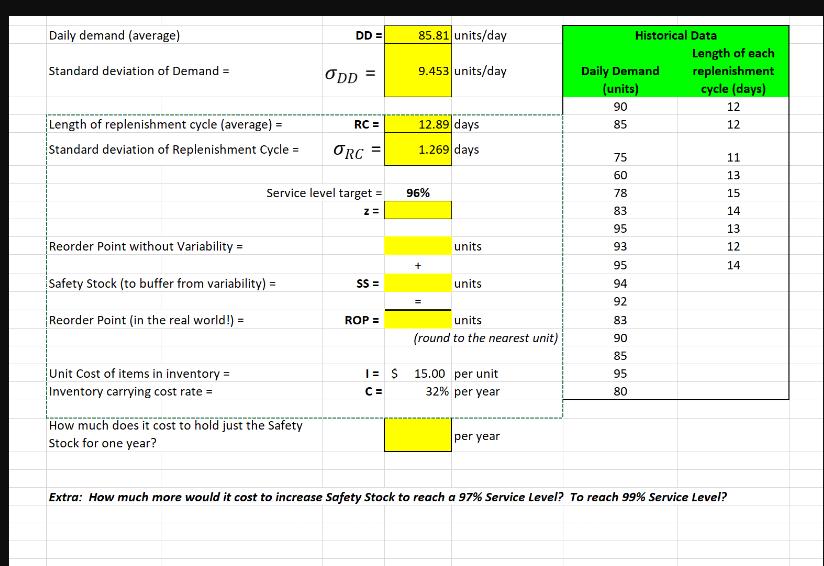
Transcribed Image Text:
Daily demand (average) Standard deviation of Demand = Length of replenishment cycle (average) = Standard deviation of Replenishment Cycle = Reorder Point without Variability= Safety Stock (to buffer from variability) = Reorder Point (in the real world!) = Unit Cost of items in inventory = Inventory carrying cost rate = DD = How much does it cost to hold just the Safety Stock for one year? ODD = RC = Service level target= Z= ORC SS= ROP = I= S C= 85.81 units/day 9.453 units/day 12.89 days 1.269 days 96% units units units (round to the nearest unit) 15.00 per unit 32% per year per year Daily Demand (units) 90 85 75 60 78 83 95 93 Historical Data 95 94 92 83 90 85 95 80 Length of each replenishment cycle (days) 12 12 11 13 15 14 13 12 14 Extra: How much more would it cost to increase Safety Stock to reach a 97% Service Level? To reach 99% Service Level? Daily demand (average) Standard deviation of Demand = Length of replenishment cycle (average) = Standard deviation of Replenishment Cycle = Reorder Point without Variability= Safety Stock (to buffer from variability) = Reorder Point (in the real world!) = Unit Cost of items in inventory = Inventory carrying cost rate = DD = How much does it cost to hold just the Safety Stock for one year? ODD = RC = Service level target= Z= ORC SS= ROP = I= S C= 85.81 units/day 9.453 units/day 12.89 days 1.269 days 96% units units units (round to the nearest unit) 15.00 per unit 32% per year per year Daily Demand (units) 90 85 75 60 78 83 95 93 Historical Data 95 94 92 83 90 85 95 80 Length of each replenishment cycle (days) 12 12 11 13 15 14 13 12 14 Extra: How much more would it cost to increase Safety Stock to reach a 97% Service Level? To reach 99% Service Level?
Expert Answer:
Answer rating: 100% (QA)
Given Daily demand average DD 8581 unitsday Standard deviation of Demand DD 9453 unitsda... View the full answer

Related Book For 

Quantitative Methods for Business
ISBN: 978-0324651751
11th Edition
Authors: David Anderson, Dennis Sweeney, Thomas Williams, Jeffrey cam
Posted Date:
Students also viewed these general management questions
-
Molander Corporation is a distributor of a sun umbrella used at resort hotels. Data concerning the next month s budget appear below: Selling price per unit $ 2 4 Variable expense per unit $ 1 5 Fixed...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
A researcher wanted to find out if there was difference between older movie goers and younger movie goers with respect to their estimates of a successful actors income. The researcher first...
-
Find each limit in Problems 3760. Note that LHpitals rule does not apply to every problem, and some problems will require more than one application of LHpitals rule. lim x 3 x + 3xx - 3 X x + 6x + 9
-
A lot of 100 semiconductor chips contains 20 that are defective. (a) Two are selected, at random, without replacement, from the lot. Determine the probability that the second chip selected is...
-
The electron in a certain hydrogen atom has an angular momentum of 8.948 x 10-34 J.s. What is the largest possible magnitude for the z component of the angular momentum of this electron? For...
-
Waves on the surface of a tank are observed to travel at a speed of \(2 \mathrm{~m} / \mathrm{s}\). How fast would these waves travel if (a) the tank were in an elevator accelerating downward at a...
-
On January 2, 2009, Weston Company acquired 20% of the 200,000 shares of outstanding common stock of Startile Corp. for $18 per share. The purchase price was equal to Startile's underlying book...
-
Analyze the integration of digital technologies, such as process simulation, computational fluid dynamics (CFD), and advanced control systems, in achieving process intensification, focusing on...
-
1. Personality is a relatively stable set of traits that aids in explaining and predicting individual behavior. What are some of Mark Cuban's traits that can explain his behavior during Maverick...
-
What is the labyrinth that the USA and China entangled with each other in ensuring its status of Superpower which would be put forward in the next two decades and how Asian and African continents can...
-
An adjusted trial balance is a list of accounts and balances prepared after recording and posting adjusting entries. Financial statements are often prepared from the adjusted trial balance. Revenue...
-
Consider the model problem below, find A and R matrices, and b and r vectors. Then find the solution: -(xu'(x))' = 4x x EI = [0, 3] xu' (0)=0; u(L)=5; ko=0; k=1 u(0)=5; 9L=5; 90=1;
-
TWC issues bullet bonds at 8% , with a ten-year maturity, company will recognize holders of securities in amount of $ 1,000 . With this information assume that changes and valuation will occur at end...
-
Which strategy utilizes the concept of "think globally, act locally"?
-
Observe the balance sheet give below of Jindal steel and power limited and give an Explanation by observing the changes happening in: 1. Fixed assets 2. depreciation 3. Cash equivalents 4. loans and...
-
When is the power of suppliers high?
-
What are the key elements of a system investigation report?
-
The Northshore Bank is working to develop an efficient work schedule for full-time and part-time tellers. The schedule must provide for efficient operation of the bank including adequate customer...
-
Davison Construction Company is building a luxury lakefront home in the Finger Lakes region of New York. Coordination of the architect and subcontractors will require a major effort to meet the...
-
Heller Manufacturing has two production facilities that manufacture baseball gloves. Production costs at the two facilities differ because of varying labor rates, local property taxes, type of...
-
A liquid enters an adiabatic throttling valve, illustrated in Figure 6-4, at T = 80F and P = 5 atm, at which conditions it has V-0.100 L/mol. The exiting liquid has P = 0.5 atm. For this steady-state...
-
100 kg/min of liquid nitrogen is produced by the steady-state process shown in Figure 5-17. 1. Nitrogen enters the process at P = 1 bar and T = 300 K. 2. The nitrogen is cooled in a heat exchanger...
-
Revisit the two processes described in Examples 4-2 and 4-3. Applying Equation 4.14, calculate the changes in entropy for the gas, the surroundings, and the universe for both processes.

Study smarter with the SolutionInn App


