Give formulas for compounds A and B: CH,COOH HBr 5a-Cholest-2-ene A (an epoxide) B (Hint: B...
Fantastic news! We've Found the answer you've been seeking!
Question:
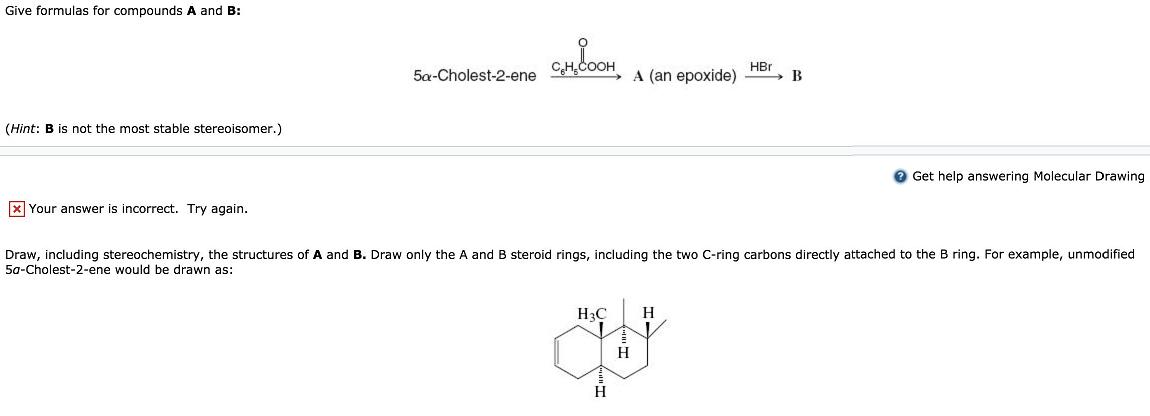
Transcribed Image Text:
Give formulas for compounds A and B: CH,COOH HBr 5a-Cholest-2-ene A (an epoxide) B (Hint: B is not the most stable stereoisomer.) O Get help answering Molecular Drawing x Your answer is incorrect. Try again. Draw, including stereochemistry, the structures of A and B. Draw only the A and B steroid rings, including the two C-ring carbons directly attached to the B ring. For example, unmodified 5a-Cholest-2-ene would be drawn as: H3C H H H Give formulas for compounds A and B: CH,COOH HBr 5a-Cholest-2-ene A (an epoxide) B (Hint: B is not the most stable stereoisomer.) O Get help answering Molecular Drawing x Your answer is incorrect. Try again. Draw, including stereochemistry, the structures of A and B. Draw only the A and B steroid rings, including the two C-ring carbons directly attached to the B ring. For example, unmodified 5a-Cholest-2-ene would be drawn as: H3C H H H
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A binary ideal-gas mixture of A and B undergoes an isothermal, isobaric separation at To, the infinite surroundings temperature. Starting with Eq. (4), Table 2.1, derive an equation for the minimum...
-
The following data apply to the binary system of A and B: Melting point of pure A = 1050C Melting point of pure B = 1900C At 1250C, the a solid solution (containing 50% of B), the B solid solution...
-
The ________ of A and B consists of all elements in either A or B or both.
-
Outline suggestions to make observation a useful, reliable requirements elicitation technique.
-
An accountant wishes to predict direct labor cost (y) on the basis of the batch size (x) of a product produced in a job shop. Data for 12 production runs are given in the table below, along with the...
-
What impact do direct and indirect bankruptcy costs have on a firms capital structure decision?
-
The Drucker Institute ranks corporations for managerial effectiveness based on a composite score derived from the following five factors: customer satisfaction, employee engagement and development,...
-
Unlevered Beta Counts Accounting has a beta of 1.15. The tax rate is 40%, and Counts is financed with 20% debt. What is Counts unlevered beta?
-
A customer has a concern of slow cranking only on a hot restart. During the visual inspection, vou also notice the insulation on the negative cable is miched. What may cause this problem? a. The...
-
On December 1, 2020, Papadopoulos Seasonings had the following account balances. During December, the company completed the following transactions. Dec. 7 Received 3,600 cash from customers in...
-
Highlight different ethical issues in Education. How can we reform our youth for such ethical consideration in the teaching-learning process and in research as well?
-
A taxpayer has $5,000 in investment expenses connected to portfolio income and is in the 8% tax bracket. Assuming the taxpayer itemizes their California return, and that these expenses are the only...
-
How can organizations foster a culture of diversity, equity, and inclusion within teams, proactively addressing systemic biases and promoting equitable access to opportunities for professional growth...
-
How might the implementation of continuous feedback mechanisms and performance metrics, coupled with regular reflection sessions and developmental coaching interventions, facilitate ongoing learning...
-
What methodologies and approaches can be employed to mitigate interpersonal conflicts and enhance interpersonal cohesion within teams, thereby fortifying resilience and bolstering performance amidst...
-
what ways can synergistic team dynamics be cultivated within organizational frameworks to augment operational efficacy and foster a culture of collaborative excellence? Explain
-
Generative AI apps like ChatGPT and Stable Diffusion have rapidly transformed many professions in only a few months of release. Explain the impact of one such tool on one industry. What is the major...
-
MgO prevents premature evaporation of Al in a furnace by maintaining the aluminum as Al2O3. Another type of matrix modifier prevents loss of signal from the atom X that readily forms the molecular...
-
Show how you could prepare each of the following compounds from propene and any necessary organic or inorganic reagents: (a) Allyl bromide (e) 1,2,3-Tribromopropane (b) 1,2-Dibromopropane (f) Allyl...
-
One of the principal substances obtained from archaea (one of the oldest forms of life on earth) is derived from a 40-carbon diol. Given the fact that this diol is optically active, is it compound A...
-
Provide a reasonable explanation for each of the following observations: (a) 4-Methylpiperidine has a higher boiling point than N-methylpiperidine. (b) Two isomeric quaternary ammonium salts are...
-
(a) Consider the flash separation process shown in Figure 7.1. If using ASPEN PLUS, solve all three cases using the MIXER, FLASH2, FSPLIT, and PUMP modules and the RK-SOAVE option set for...
-
As discussed in Example 6.7, toluene \(\left(\mathrm{C}_{7} \mathrm{H}_{8} ight)\) is to be converted thermally to benzene \(\left(\mathrm{C}_{6} \mathrm{H}_{6} ight)\) in a hydrodealkylation...
-
As discussed in Example 6.7, the following stream at \(100^{\circ} \mathrm{F}\) and 484 psia is to be separated by two distillation columns into the Products \(1-3\) in the following table. Two...

Study smarter with the SolutionInn App


