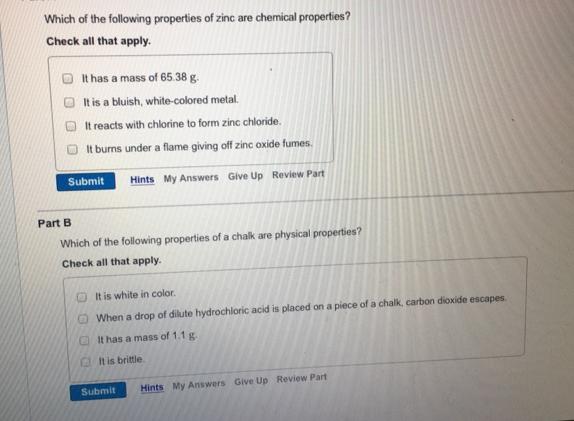
Which of the following properties of zinc are chemical properties? Check all that apply. It has...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Which of the following properties of zinc are chemical properties? Check all that apply. It has a mass of 65.38 g. It is a bluish, white-colored metal. It reacts with chlorine to form zinc chloride. It burns under a flame giving off zinc oxide fumes. Submit Hints My Answers Give Up Review Part Part B Which of the following properties of a chalk are physical properties? Check all that apply. It is white in color. When a drop of dilute hydrochloric acid is placed on a piece of a chalk, carbon dioxide escapes. It has a mass of 1.1 g. It is brittle Submit Hints My Answers Give Up Review Part Which of the following properties of zinc are chemical properties? Check all that apply. It has a mass of 65.38 g. It is a bluish, white-colored metal. It reacts with chlorine to form zinc chloride. It burns under a flame giving off zinc oxide fumes. Submit Hints My Answers Give Up Review Part Part B Which of the following properties of a chalk are physical properties? Check all that apply. It is white in color. When a drop of dilute hydrochloric acid is placed on a piece of a chalk, carbon dioxide escapes. It has a mass of 1.1 g. It is brittle Submit Hints My Answers Give Up Review Part
Expert Answer:
Answer rating: 100% (QA)
The detailed answer for the above question is provided below 1 The following properties of zinc are ... View the full answer

Related Book For 

Equity Asset Valuation
ISBN: 978-0470571439
2nd Edition
Authors: Jerald E. Pinto, Elaine Henry, Thomas R. Robinson, John D. Stowe, Abby Cohen
Posted Date:
Students also viewed these chemistry questions
-
Which of the following properties indicates very strong intermolecular forces in a liquid? (a) Very low surface tension, (b) Very low critical temperature, (c) Very low boiling point, (d) Very low...
-
A flask containing dilute hydrochloric acid was placed on a digital balance. An excess of limestone chippings was added to this acid, a plug of cotton wool was placed in the neck of the flask and the...
-
When resistors are connected in series, which of the following would be the same for each resistor: potential difference, current, power?
-
Solve the inequality. Write the solution in interval notation. |15 = x < 7
-
Fast Service Truck Lines uses the Ford Super Duty F-750 exclusively. Management made a study of the maintenance costs and determined the number of miles traveled during the year followed the normal...
-
Horizontal forces of 3 N and 4 N act at right angles on a block of mass 5 kg. Show that the resulting acceleration is 1 m/s 2 .
-
What is the purpose of timelines in an investigation?
-
Placid Lake Corporation acquired 80 percent of the outstanding voting stock of Scenic, Inc., on January 1, 2014, when Scenic had a net book value of $400,000. Any excess fair value was assigned to...
-
First Draw an ER Model please and then covert it into relational model seperate figures required thank you Each cinema is identified by its name and has its residency at an address which consists of...
-
Submag, Inc., uses direct-mail promotion to sell magazine subscriptions. Magazine publishers pay Submag $3.12 for each new subscription. Submags costs include the expenses of printing, addressing,...
-
The isolation of a short polypeptide yields 230 kg of butanol extract containing 0.081 wt% peptide. We want to strip this extract with initially peptide-free saline solution at pH 9.0 to yield a...
-
Consider the following statement: Vinteger n, if n is even then n is even . Which of the following are equivalent ways of expressing this state- ment? (a). All integers have even squares and are...
-
Prior to beginning work on this discussion forum, read Chapter 1 of your textbook. On the Internet, find the most recent quarterly sales, comparable sales, financial report, or any other data set for...
-
Case Study Link: https://www.coursehero.com/u/file/87901915/1617954788513-1617917843658-Assignment-2257pdf/?justUnlocked=1#/question Please refer the case study to answer the following questions:...
-
Question 8: What can you say for certain? Part 1 (a) A must be true (b) B must be true (c) A must be false (d) B must be false (e) None of these Select all possible options that apply. ? Mystery A U...
-
Marc wants to invest in either LOP or ABC shares.He found out that last year LOP paid $0.50 in dividends while ABC paid dividends of $1.25 last year.LOP dividends is anticipated to grow at a rate of...
-
ABC Co. is preparing its September 30, 20x1 bank reconciliation. Relevant information is shown below: Balance per books Balance per bank statement Collection of note receivable by bank fincluding...
-
AB CORPORATION ISSUED THE FOLLOWING 850 COMMON STOCKS PAR VALUE P100 750 PARTICIPATING PREFERRED STOCKS PAR VALUE P100 AT 3% AB CORPORATION DECLARED P100,000.00 DIVIDEND IN 2022.
-
The text defined intrinsic value as the value of an asset given a hypothetically complete understanding of the assets investment characteristics. Discuss why hypothetically is included in the...
-
Watson Dunn is planning to value BCC Corporation, a provider of a variety of industrial metals and minerals. Dunn uses a single - stage FCFF approach. The financial information Dunn has assembled for...
-
An analyst plans to use P/E and the method of comparables as a basis for recommending purchasing shares of one of two peer - group companies in the business of manufacturing personal digital...
-
How much money would have to be deposited today to accumulate a. $10,000 after 6 years if the investment earns 5%/year compounded annually. b. $6,500 after 4 years if the investment earns 8...
-
What will be the amount accumulated by each of the following present investments? a. $3,000 invested for 7 years at 14 percent compounded annually b. $1,600 invested for 17 years at 12% compounded...
-
On August 1, 1958, first-class postage for a 1-ounce envelope was 4. On August 1, 2007, a first-class stamp for the same envelope cost 41. What was the annual compound increase in the cost of...

Study smarter with the SolutionInn App


