Even liquids and solids are slightly compressible. A common phenomenological form for the volume as a...
Fantastic news! We've Found the answer you've been seeking!
Question:
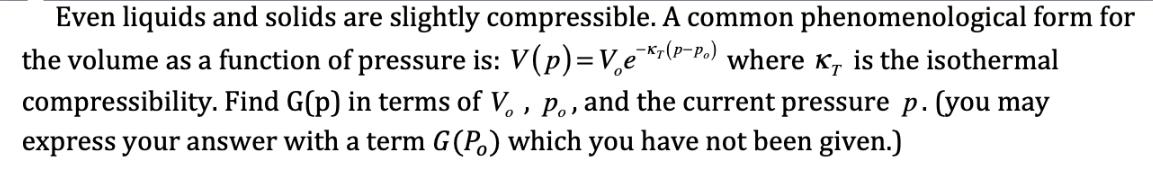

Transcribed Image Text:
Even liquids and solids are slightly compressible. A common phenomenological form for the volume as a function of pressure is: V(p)= Ver(P-Po) where K, is the isothermal compressibility. Find G(p) in terms of V., P., , and the current pressure p. (you may express your answer with a term G (P) which you have not been given.) KT for liquid water is 5.0×10 atm ¹. Using the form for the compressibility given in the last problem, how much pressure needs to be applied to compress one liter of water to a volume of .99 liters? Even liquids and solids are slightly compressible. A common phenomenological form for the volume as a function of pressure is: V(p)= Ver(P-Po) where K, is the isothermal compressibility. Find G(p) in terms of V., P., , and the current pressure p. (you may express your answer with a term G (P) which you have not been given.) KT for liquid water is 5.0×10 atm ¹. Using the form for the compressibility given in the last problem, how much pressure needs to be applied to compress one liter of water to a volume of .99 liters?
Expert Answer:
Answer rating: 100% (QA)
The first question describes the relationship between volume and pressure using an exponential funct... View the full answer

Related Book For 

Posted Date:
Students also viewed these physics questions
-
Draw a free body diagram of an individual performing calfraises, rising onto the balls of their feet and then lowering theirheels. Draw a free body diagram of an individual performing overheadtriceps...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
There are two major producers of corncob pipes in the world. Suppose that the inverse demand function for corncob pipes is described by p = 120 - 4g where g is total industry output and suppose that...
-
To the left of each account listed on the trial balance, indicate the financial statement that will include the account: income statement (IS), statement of retained earnings (RE), or balance sheet...
-
Twenty-three overweight male volunteers are randomly assigned to three different treatment programs designed to produce a weight loss by focusing on either diet, exercise, or the modification of...
-
Consider the multiple regression model fit to the house price data in Problem 3.7. Problem 3.7 Consider the house price data in Table B.4. a. Construct a normal probability plot of the residuals....
-
Data pertaining to the current position of Boole Company are as follows: Cash ................ $240,000 Temporary investments ......... 120,000 Accounts and notes receivable (net) ... 360,000...
-
Using the template proposed below, write a Matlab function fwkinrpp that implements the forward kinematics for the RPP manipulator pictured in Figure 3. function T fwkinrpp (q) end % q is a 3x1...
-
Ramada Company produces one golf cart model. A partially complete table of company costs follows: Required: 1. Complete the table. 2. Ramada sells its carts for $1,200 each. Prepare a contribution...
-
Explain in details the difference between managing a software project and other projects.
-
A \(0.10-\mathrm{kg}\) pendulum swings inside a box that contains \(1.0 \times 10^{23}\) nitrogen molecules. The initial speed of the pendulum is \(0.80 \mathrm{~m} / \mathrm{s}\), and it comes to...
-
A box is divided into four equal-sized quadrants. Each quadrant contains gaseous hexane \(\left(\mathrm{C}_{6} \mathrm{H}_{14} ight)\), which we treat as monatomic. The partition that separates the...
-
Chamber \(\mathrm{X}\) contains monatomic ideal gas \(\mathrm{X}\). The gas particles have a root-mean-square speed of \(42 \mathrm{~m} / \mathrm{s}\). Chamber Y, identical to chamber X, contains a...
-
A sealed \(1.50-\mathrm{L}\) chamber filled with helium gas initially at \(20^{\circ} \mathrm{C}\) and \(1.00 \mathrm{~atm}\) is heated until the gas temperature is \(232^{\circ} \mathrm{C}\). (a)...
-
The atoms of an element can come in different forms, depending on the number of neutrons in the nucleus. These different forms are called isotopes of the element. The two most common isotopes of...
-
Ayayai Corporation had the following activities in 2020. 1. Sold land for $206,000. 2. Purchased an FV-NI investment in common shares for $14,900. 3. Purchased inventory for $863,000 with cash. 4....
-
A 2500-lbm car moving at 15 mi/h is accelerated at a constant rate of 15 ft/s 2 up to a speed of 50 mi/h. Calculate force and total time required?
-
The reactions described in Example 12-7 are to be carried out isothermally at 1000C in the reactor whose RTD is described in Example 17-4 with C A0 = C B0 = 0.05 mol/dm 3 . a. Determine the exit...
-
For the decomposition of cumene discussed in this chapter, if an adsorbing inert is present, how would you compare the initial rate as a function of total pressure when desorption is the RLS, as...
-
The irreversible reaction A B is taking place in the same porous catalyst slab shown in Figure P15-9A. The reaction is zero order in A. a. Show that the concentration profile using the symmetry B.C....
-
To test \(H_{0}: \sigma=50\) versus \(H_{1}: \sigma <50\), a random sample of size \(n=24\) is obtained from a population that is known to be normally distributed. (a) If the sample standard...
-
(a) Determine the critical values for a right-tailed test of a standard population deviation with 18 degrees of freedom at \(\alpha=0.1\) level of significance. (b) Determine the critical values for...
-
In Example 2 from Section 10.3, the quality-control engineer for M\&MMars tested whether the mean weight of fun-size Snickers was 20.1 grams. Suppose that the standard deviation of the weight of the...

Study smarter with the SolutionInn App


