Question: A rigid tank is divided by a partition into 2 equal parts. Initially, one side of the tank has 5 kg of water at
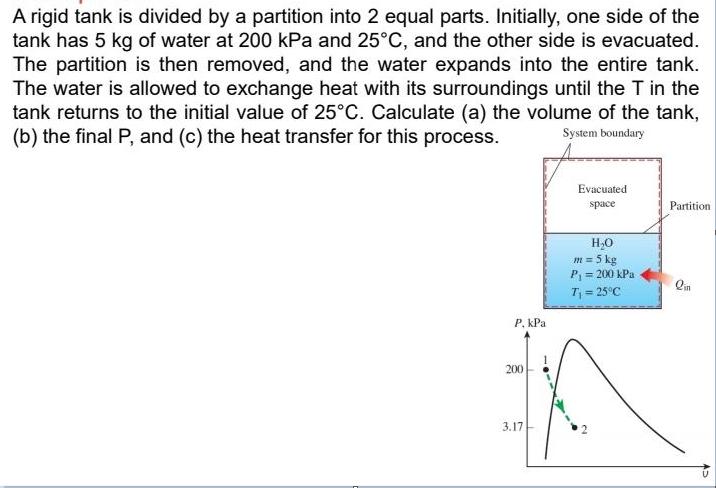
A rigid tank is divided by a partition into 2 equal parts. Initially, one side of the tank has 5 kg of water at 200 kPa and 25C, and the other side is evacuated. The partition is then removed, and the water expands into the entire tank. The water is allowed to exchange heat with its surroundings until the T in the tank returns to the initial value of 25C. Calculate (a) the volume of the tank, (b) the final P, and (c) the heat transfer for this process. System boundary Evacuated space Partition H,0 m = 5 kg P = 200 kPa T = 25C P, kPa 200 3.17
Step by Step Solution
3.45 Rating (158 Votes )
There are 3 Steps involved in it

Get step-by-step solutions from verified subject matter experts


