. 3-7. Looking only at the periodic table, write the complete electron configuration (152 252 etc.)...
Fantastic news! We've Found the answer you've been seeking!
Question:
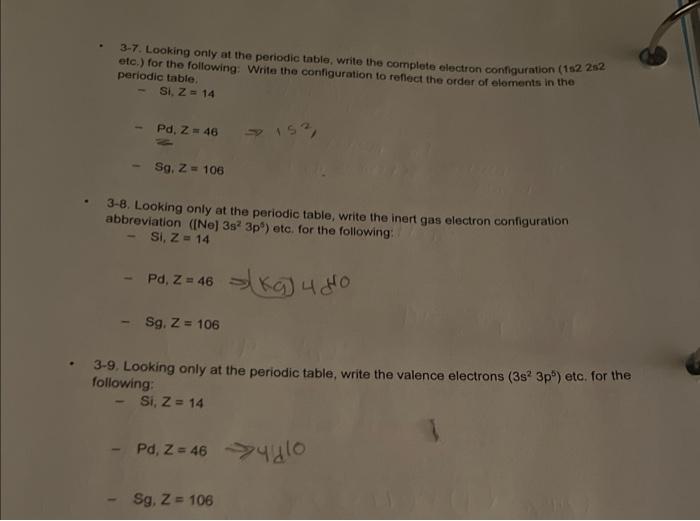
Transcribed Image Text:
. 3-7. Looking only at the periodic table, write the complete electron configuration (152 252 etc.) for the following: Write the configuration to reflect the order of elements in the periodic table. Si, Z = 14 Pd, Z = 46 Sg. Z=106 3-8. Looking only at the periodic table, write the inert gas electron configuration abbreviation ([Ne] 3s 3p) etc. for the following: Si, Z=14 - Pd, Z = 46kg) 4 (40 Sg. Z = 106 153 3-9. Looking only at the periodic table, write the valence electrons (3s² 3p) etc. for the following: Si, Z = 14 - Pd, Z=46 Sg, Z = 106 . 3-7. Looking only at the periodic table, write the complete electron configuration (152 252 etc.) for the following: Write the configuration to reflect the order of elements in the periodic table. Si, Z = 14 Pd, Z = 46 Sg. Z=106 3-8. Looking only at the periodic table, write the inert gas electron configuration abbreviation ([Ne] 3s 3p) etc. for the following: Si, Z=14 - Pd, Z = 46kg) 4 (40 Sg. Z = 106 153 3-9. Looking only at the periodic table, write the valence electrons (3s² 3p) etc. for the following: Si, Z = 14 - Pd, Z=46 Sg, Z = 106
Expert Answer:
Answer rating: 100% (QA)
Solution 37 Electronic configuration Acc to Aufbaus principle Energy levels of orbitals be d... View the full answer

Related Book For 

Valuation Measuring and managing the values of companies
ISBN: ?978-0470424704
5th edition
Authors: Mckinsey, Tim Koller, Marc Goedhart, David Wessel
Posted Date:
Students also viewed these corporate finance questions
-
Using the methodology outlined in Exhibit 6.16, determine equity cash flow for year 1. Use the growing-perpetuity formula (based on equity cash flow) to compute BrandCos equity value. Assume the cost...
-
Using the methodology outlined in Exhibit 9.10, forecast the operating items on next year's balance sheet for PartsCo. Forecast each balance sheet item as a function of revenue, except inventory and...
-
Using the methodology outlined in Exhibit 9.12, forecast the financing items on next year's balance sheet for PartsCo. Assume long-term debt remains at $215 million, no external equity is raised, and...
-
An opera glass has an objective lens of focal length +3.60 cm and a negative eyepiece of focal length -1.20 cm. How far apart must the two lenses be for the viewer to see a distant object at 25.0 cm...
-
Let X1, . . ., Xn be a random sample from a negative binomial distribution with pmf Find method of moments estimators for r and p. [Here E[X] = r(1 p) / p and E[X2] = r(1 p) (r rp + 1) / p2.]...
-
An elementary particle is launched from Earth toward the Regulus system, 77. 5 light years distant. At what speed relative to Earth must this particle travel to make this trip in \(10 \mathrm{y}\) in...
-
What items of information must be included in a notice of deposition?
-
Ken Smith, the partner in charge of the audit of Houghton Enterprises, identified the following significant deficiencies during the audit of the December 31, 2013, financial statements: 1. Controls...
-
1. Discuss briefl y the four cultural predispositions MNCs tend to have towards managing things in a global context. 2. Draw and describe the phases of cultural adjustment that an expatriate...
-
Ozone gas (O 3 , solute A) dissolved in high-purity water is commonly used in wet cleaning processes associated with semiconductor device fabrication. It is desired to produce a liquid water stream...
-
Required information [The following information applies to the questions displayed below] a. On March 22, purchased 790 shares of RPI Company stock at $13 per share. Duke's stock investment results...
-
3:01 Flight Caf prepares in-flight meals for airlines in its kitchen located next to a local airport. The company's planning budget for July appears below: Flight Caf Planning Budget For the Month...
-
On January 1, Ruby Inc. Issued 5,000 of $1,000 par value bonds with a stated rate of 6% and a 10-year maturity. Interest is payable semiannually on June 30 and December 31. Use Future Value of a...
-
How do organisms navigate complex environments and coordinate behaviors, and what are the neural and sensory mechanisms underlying these adaptive responses ?
-
I Why is SCM so important right now in our current environment of business? How can we keep from having a shortage of items in times of crisis like a pandemic?
-
Silven Industries, which manufactures and sells summer lotions and insect repellents, has decided to diversify in order to stabilize sales throughout the year. A natural area for the company to...
-
Describe briefly how language can influence a person, be they the recipient or the messenger, and explain the reason social work as a profession has chosen to take great care with the language it...
-
I frequently use NY Times and CNN and am aware of Fox News but I never use it. I visit these sites, NY Times and CNN, a few times a week whenever I have to research something or see something on...
-
What is financial engineering? When does it create value?
-
Describe the five-step approach to combining nominal and real forecasts.
-
ToyCo has working capital of $400 million, fixed assets equal to $800 million, and debt equal to $600 million. Use this data and the reorganized deferred taxes in Question 4 to create invested...
-
In April 2023, HMRC issues a notice requiring an individual to submit a tax return for the year 2022-23. The return is submitted electronically to HMRC on 8 December 2023. (a) State the date by which...
-
Calculate the personal savings allowance available in 2023-24 to a taxpayer with taxable income for the year (i.e. net income less any available personal allowance) of: (a) 20,000 (b) 37,701 (c)...
-
Calculate the 2023-24 income tax liability of a non-Scottish taxpayer with taxable income (i.e. income remaining after deducting any available personal allowance) of: (a) 11,730 (b) 15,280 (c) 30,000...

Study smarter with the SolutionInn App


