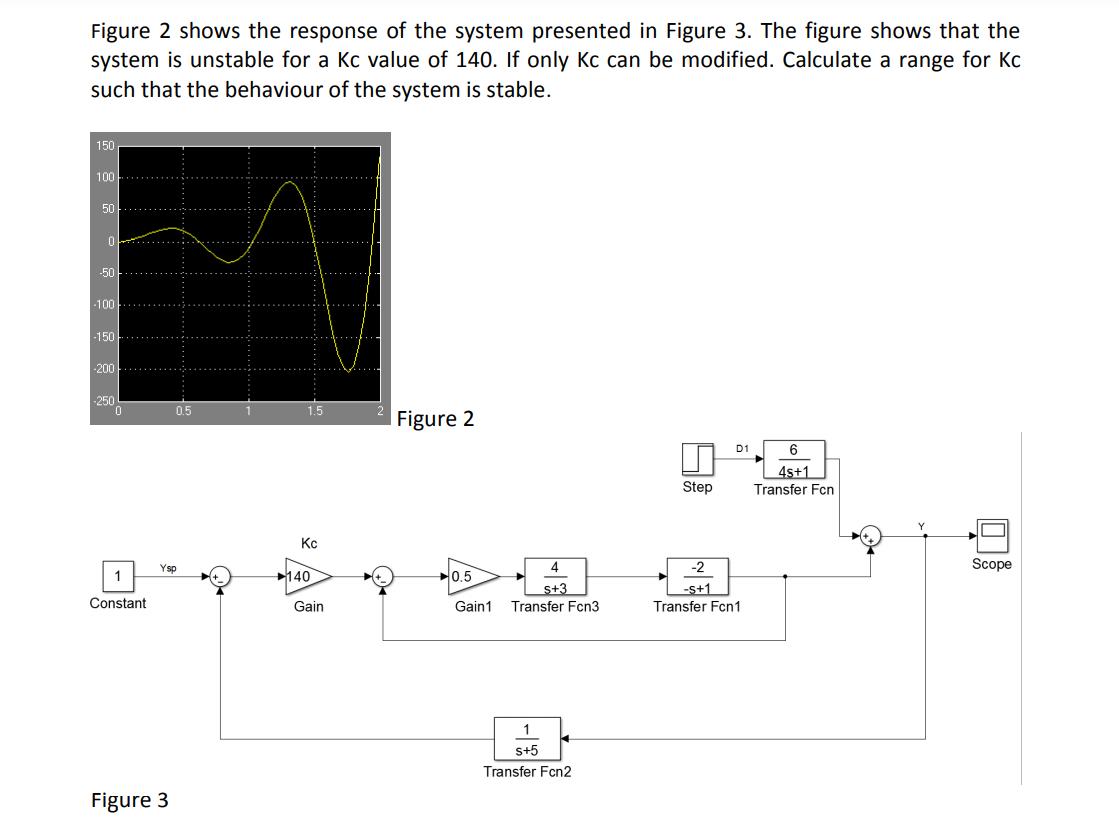
Figure 2 shows the response of the system presented in Figure 3. The figure shows that...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Figure 2 shows the response of the system presented in Figure 3. The figure shows that the system is unstable for a Kc value of 140. If only Kc can be modified. Calculate a range for Kc such that the behaviour of the system is stable. 150 100 50 -50 -100 -150 -200 -250 0.5 1.5 Figure 2 D1 6 4s+1 Transfer Fcn Step Kc Ysp 4 -2 Scope 1 140 0.5 s+3 -s+1 Constant Gain Gain1 Transfer Fcn3 Transfer Fcn1 s+5 Transfer Fcn2 Figure 3 中 Figure 2 shows the response of the system presented in Figure 3. The figure shows that the system is unstable for a Kc value of 140. If only Kc can be modified. Calculate a range for Kc such that the behaviour of the system is stable. 150 100 50 -50 -100 -150 -200 -250 0.5 1.5 Figure 2 D1 6 4s+1 Transfer Fcn Step Kc Ysp 4 -2 Scope 1 140 0.5 s+3 -s+1 Constant Gain Gain1 Transfer Fcn3 Transfer Fcn1 s+5 Transfer Fcn2 Figure 3 中
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
Figure shows the response of a system to a step input of magnitude 1000 N. The equation of motion is m + c + kx = f(t) Estimate the values of m, c, and k. 0.04 0.036 0.032 0.028 0.024 E 0.02 0.016...
-
The response of a system to a unit step change in the input (occurring at time 0) is shown in Fig. E7.11. (a) Derive a second-order plus time delay model approximation for the system. Provide values...
-
Compute the response of the system y(n) = 0.7y (n 1) -0.12y(n 2) + x(n 1) + x(n 2) to the input x(n) = nu(n). Is the system stable?
-
Suppose you are a depositor at First Street Bank. You hear a rumor that the bank has suffered serious losses on its loans. Every depositor knows that the rumor isnt true, but each thinks that most...
-
Zhang Company's financial records reveal the following at 30 June 2013. Net sales .......................................$196 000 Cost of sales ...................................$104 000 Ending...
-
The tip of a soldering iron consists of a 0.6-cm-OD copper rod, 7.6 cm long. If the tip must be 204?C, what is the required minimum temperature of the base and the heat flow, in Btu??s per hour and...
-
Discuss the theoretical bases for the four HR administrative approaches introduced in this chapter. Are such theories useful to HR professionals in their efforts to improve transactional performance?...
-
Deines Company accumulates the following data concerning a mixed cost, using miles as the activity level. Compute the variable and fixed cost elements using the high-low method. Miles Driven 8,000...
-
a. Performed $8,200 of services on account. b. Collected $5,600 cash on accounts receivable. c. Paid $1,450 cash in advance for an insurance policy. d. Paid $400 on accounts payable. e. Recorded the...
-
If an amino acid is found at the exact same location in the alignment of two homologous proteins, that amino acid is said to be "absolutely conserved." An amino acid type can also be conserved at a...
-
An aqueous solution of methanol is being separated by distillation in a column. Methanol (A). which is the more volatile component, moves from liquid phase to the vapor phase while water (B), the...
-
Explain how each relates to the NASW Code of Ethics and how case management ethics is rooted in social work. How would an ethical dilemma be dealt with by a social worker with a Christian worldview?
-
Explain once upon a time in Arkansas and rose law firm billing records
-
Sunset & Vine Wine Bar, a seasonal business, collected the following information for specific months of operation Month Hours Open Amount 280 Server labor costs September $5,600 February 2,400...
-
Consider the following function. 1 f(x)= = 2 (x-1) Identify the shape of the basic function
-
An accrual basis corporate taxpayer, Pale Tourist, Inc., began business operations on September 1. It incurred the following expenses in connection with organizing the corporation: (1) legal fees for...
-
On October 1, Ebony Ernst organized Ernst Consulting; on October 3, the owner contributed $84,310 in assets in exchange for its common stock to launch the business. On October 31, the companys...
-
Which of the ocean zones shown would be home to each of the following organisms: lobster, coral, mussel, porpoise, and dragonfish? For those organisms you identify as living in the pelagic...
-
Figure shows a system for controlling the angular position of a load, such as an antenna. Figure shows the block diagram for PD control of this system using a field-controlled motor. Use the...
-
A rigid container has a volume of 20 ft3. The air inside is initially at 70"F. Find the pneumatic capacitance of the container for an isothermal process.
-
Figure shows an engine valve driven by an overhead camshaft. The rocker arm pivots about the fixed point O. For particular values of the parameters shown, the valve displacement x(t) satisfies the...
-
If you let go of a helium balloon, it quickly rises. As it rises, the balloon gets larger and larger until it pops. Why does the balloon expand as it rises?
-
Elephant seals do deep dives to forage for food. The energy used by the seals in diving depends on their body composition; for example, fatter seals use less energy to swim to the surface at the end...
-
Early airshipscraft that relied on large volumes of low density gas to rise from the groundwere filled with hydrogen, but safety concerns necessitated a switch to helium. When an airship made the...

Study smarter with the SolutionInn App