Question: Figure below is experimental data on the solubility, primary nucleation threshold (PNT) and secondary nucleation threshold (SNT) for a protein, alpha amylase. I wish
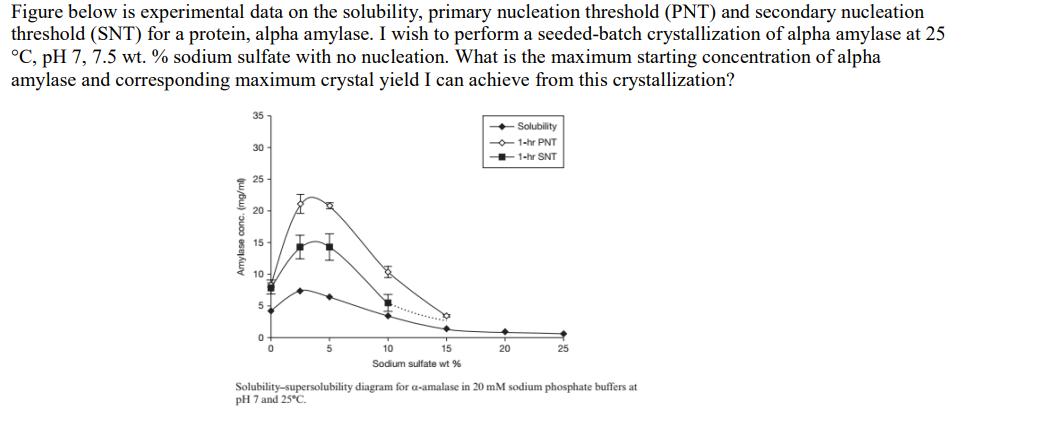
Figure below is experimental data on the solubility, primary nucleation threshold (PNT) and secondary nucleation threshold (SNT) for a protein, alpha amylase. I wish to perform a seeded-batch crystallization of alpha amylase at 25 C, pH 7, 7.5 wt. % sodium sulfate with no nucleation. What is the maximum starting concentration of alpha amylase and corresponding maximum crystal yield I can achieve from this crystallization? 35 + Solubility -1-hr PNT + 1-hr SNT 30 25 20 15 10 5 10 15 20 25 Sodium sulfate wt % Solubility-supersolubility diagram for a-amalase in 20 mM sodium phosphate buffers at pH 7 and 25C. ju Bu) ouoo asejkuy
Step by Step Solution
3.45 Rating (158 Votes )
There are 3 Steps involved in it
35 Solubility 1hr PNT 30 1hr SNT 25 20 15 10 10 15 20 25 Sodium sulfate wt Solubili... View full answer

Get step-by-step solutions from verified subject matter experts


