For which of the following is hydrogen bonding NOT a factor? O H0 OCH4 C3H7OH ONH3...
Fantastic news! We've Found the answer you've been seeking!
Question:
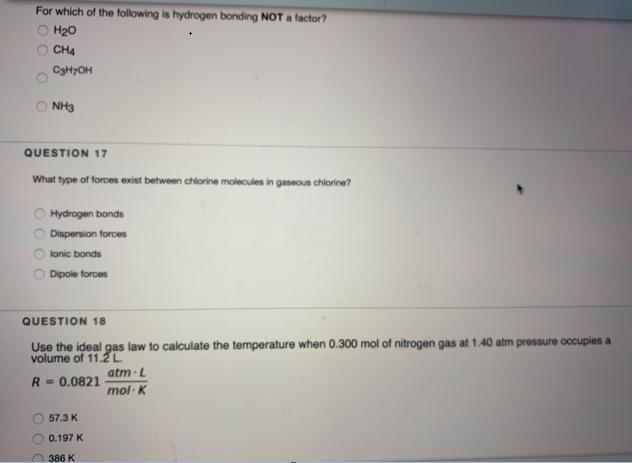
Transcribed Image Text:
For which of the following is hydrogen bonding NOT a factor? O H₂0 OCH4 C3H7OH ONH3 QUESTION 17 What type of forces exist between chlorine molecules in gaseous chlorine? 000 Hydrogen bonds Dispersion forces lonic bonds Dipole forces QUESTION 18 Use the ideal gas law to calculate the temperature when 0.300 mol of nitrogen gas at 1.40 atm pressure occupies a volume of 11.2 L atm L R = 0.0821 mol-K 57.3 K 0.197 K 386 K For which of the following is hydrogen bonding NOT a factor? O H₂0 OCH4 C3H7OH ONH3 QUESTION 17 What type of forces exist between chlorine molecules in gaseous chlorine? 000 Hydrogen bonds Dispersion forces lonic bonds Dipole forces QUESTION 18 Use the ideal gas law to calculate the temperature when 0.300 mol of nitrogen gas at 1.40 atm pressure occupies a volume of 11.2 L atm L R = 0.0821 mol-K 57.3 K 0.197 K 386 K
Expert Answer:
Answer rating: 100% (QA)
As Solution 16 we know that for hydrogen bonding any of FO N atoms ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Which of the following is not required by generally accepted auditing standards? a. Written representations. b. Attorney letter. c. Management letter. d. Engagement letter.
-
Which of the following is not an internal audit objective designed to add value to a purchas-ing department? a. A review of the bidding process indicates that a vendor company may be operating under...
-
Which of the following is considered different and more limited in objectives than the others? a. Operational auditing. b. Performance auditing. c. Management auditing. d. Financial statement...
-
1.3 WANEFMAC7 1.3.007. Find the equation of the given linear function. \[ f(x)= \] WANEFMAC7 1.3.015. Find the slope of the given line, if it is defined. (If an answer is undefined, enter UNDEFINED.)...
-
An assembly line must be designed to produce 50 packages per hour. The following data in Table 10-17 give the necessary information. (a) Draw a precedence diagram. (b) Compute the cycle time (in...
-
A battery of cylindrical hollow-fiber membranes is operated at steady state to concentrate a bacterial suspension from a fermenter. 350 kg/min fermenter broth is pumped through a stack of...
-
Refer to Googles financial statements in Appendix A to compute its equity ratio as of December 31, 2015, and December 31, 2014. Data From Google Financial Statement Appendix A Google Inc....
-
Your division is considering two projects. Its WACC is 10%, and the projects? after-tax cash flows (in millions of dollars) would be as follows: a. Calculate the projects? NPVs, IRRs, MIRRs, regular...
-
Topic : SQL DDL & DML Purpose The purpose of this assignment is to help you learn more about SQL. Brief Introduction SQL is a domain-specific language used in programming and designed for managing...
-
The data file agstrat.dat also contains information on other variables. For each of the following quantities, plot the data, and estimate the population mean for that variable along with its standard...
-
Garlic gravy manufacturer presents the following budget showing units (jars) of gravy to be produced for the upcoming months: Units to be produced. April May 82.000 80.000 407,500 grams 399,500 grams...
-
A kickback scheme has been discovered in a warehouse company that purchases office supplies for resale. The perpetrator is the purchasing agent. The agent personally benefited from receiving around...
-
Music lessons for your child will cost $1,265 per year. If you get a part-time job that pays $6.05 per hour, how many hours will you have to work in a year to pay for the music lessons?
-
Ben opened a retail store on 1 April 2017. He introduced the following into the business. Inventory 15 200 Shop fittings 14 300 Cash 17 900 (of which $17 400 was paid into a business bank account) On...
-
Prepare the finance department's budget for next year. He thinks expenses will be about $120,000, but decides to add a cushion of $80,000, for a total submitted budget of $200,000.
-
Prepare the required March 31st and June 30th journal entries. You are only completing the payroll and vacation accrual portion of the payroll entries and cash can be credited to account for actual...
-
(a): Let r be a real number such that r 1. Use induction to prove the geometric series formula: n arn i=0 = a - arn+1 1-r (b): Define an, recursively, by ao = 1 and an = 1 + Eda,. Show, using strong...
-
If the jobs displayed in Table 18.24 are processed using the earliestdue-date rule, what would be the lateness of job C? TABLE 18.24 Processing Times and Due Dates for Five Jobs Job C D E...
-
Describe the change in hybridization (if any) of the Al atom in the following reaction: AICH + Cl- AICI
-
An electron and a positron are accelerated to nearly the speed of light before colliding in a particle accelerator. The ensuing collision produces an exotic particle having a mass many times that of...
-
Consider the following compounds: Experimentally, phenol is found to be a stronger acid than methanol. Explain this difference in terms of the structures of the conjugate bases. phenol methanol
-
True or False: Engineers seldom have an opportunity to influence the recovery period for expenditures.
-
A lumber company purchases and installs a wood chipper for \(\$ 200,000\). The chipper is classified as MACRS 7-year property. Its useful life is 10 years. The estimated salvage value at the end of...
-
True or False: Straight-line depreciation is the most popular depreciation method used in financial reporting.

Study smarter with the SolutionInn App


