Given that the concentration of hydronium ions in a solution with pH of 2 is 0.19%,...
Fantastic news! We've Found the answer you've been seeking!
Question:

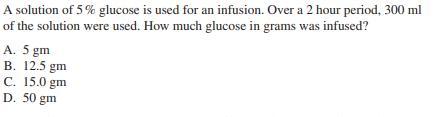
Transcribed Image Text:
Given that the concentration of hydronium ions in a solution with pH of 2 is 0.19%, what would be the concentration of hydronium ions in a solution with pH of 3? A. 0.13% B. 0.29% C. 0.019% D. 1.9% A solution of 5% glucose is used for an infusion. Over a 2 hour period, 300 ml of the solution were used. How much glucose in grams was infused? A. 5 gm B. 12.5 gm C. 15.0 gm D. 50 gm Given that the concentration of hydronium ions in a solution with pH of 2 is 0.19%, what would be the concentration of hydronium ions in a solution with pH of 3? A. 0.13% B. 0.29% C. 0.019% D. 1.9% A solution of 5% glucose is used for an infusion. Over a 2 hour period, 300 ml of the solution were used. How much glucose in grams was infused? A. 5 gm B. 12.5 gm C. 15.0 gm D. 50 gm
Expert Answer:
Answer rating: 100% (QA)
The detailed answer for the above question is provided below Part 1 Hydronium ion concentration Firs... View the full answer

Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Posted Date:
Students also viewed these medical sciences questions
-
What are some concepts of social psychology, if so if self-identity one concept of social psychology? 2.What is critical social psychology?
-
What is the concentration of hydronium ions in a solution that has a pH of 3? Why is such a solution impossible to prepare?
-
Sometimes the pH must be converted into the hydronium ion concentration. The quickest way to find the hydronium ion concentration in a solution is to use a pH meter to measure the pH and then...
-
Do economists have any particular expertise at making normative arguments? In other words, they have expertise at making positive statements (i.e., what will happen) about some economic policy, for...
-
Autotech Manufacturing is engaged in the production of replacement parts for automobiles. One plant specializes in the production of two parts: Part #127 and Part #234. Part #127 produced the highest...
-
In rolling a die, suppose that we get the first Six in the 7th trial and in doing it again we get it in the 6th trial. Estimate the probability p of getting a Six in rolling that die once.
-
What/who is the source of each initiative? lo1
-
Graphics Service Co. purchased a new color copier at the beginning of 2013 for $47,000. The copier is expected to have a five-year useful life and a $7,000 salvage value . The expected copy...
-
eBook Problem Walk-Through Weston Corporation just paid a dividend of $1.25 a share (i.e., D0 = $1.25). The dividend is expected to grow 11% a year for the next 3 years and then at 4% a year...
-
The Clampett Oil Company has a tanker truck that it uses to deliver fuel to customers. The tanker has five different storage compartments with capacities to hold 2,500, 2,000, 1,500, 1,800 and 2,300...
-
b . Why, when CUER exceeded TER, was this considered a deficiency in operation of the control
-
Given the following memory status below, compute how much does it cost to compact holes together with the following compaction strategies if 1 kbyte of movement costs 50 centavos. 0. OS OS OS OS OS...
-
ITG Pte Ltd ("ITG") is a company specialising in air-conditioner maintenance and servicing. It makes adjusting and closing entries every 31 December, which is the company's financial year-end. Unless...
-
(20 points) We know that when we have a graph with negative edge costs, Dijkstra's algo rithm is not guaranteed to work. (a) Does Dijkstra's algorithm ever work when some of the edge costs are...
-
Create a new user called cis605_usr. Use Master. assign a password of abcd, set check_policy to off and check_expiration to off (Why set these two to off?). Execute the sp_addsrvrolemember to add the...
-
Salmone Company reported the following purchases and sales of its only product. Salmone uses a periodic inventory system. Determine the cost assigned to the ending inventory using FIFO. Date Units...
-
Please assist with Part 2 and show work Splish Company uses the gross profit method to estimate inventory for monthly reporting purposes. Presented below is information for the month of May....
-
The outer loop controls the number of students. Note that the inner loop of this program is always executed exactly three times, once for each day of the long weekend. Modify the code so that the...
-
Who has the shorter wavelength when running at the same speed: a person weighing 60 kg or a person weighing 80 kg? Explain your reasoning.
-
(a) A flask of volume 350. mL contains 0.1500 mol Ar at 24 C. What is the pressure of the gas in kilopascals? (b) You are told that 23.9 mg of bromine trifluoride exerts a pressure of 10.0 Torr at...
-
The chemists who synthesized the pentamethylcyclopentadienyl cation (see Exercise 11.11) found that its p-electrons are found in one double bond and a p-orbital that is delocalized across the three...
-
At the end of May, the sales journal of Mountain View appears as follows. Sales Journal Date Account Debited Invoice Number PR Cost of Goods Sold Dr. Inventory Cr. Accounts Receivable Dr. Sales Cr....
-
Prepare headings for a cash disbursements journal like the one in Exhibit E-A.4. Journalize the April transactions from Exercise E-10 that should be recorded in the cash disbursements journal...
-
Houst Pharmacy uses the following journals: sales journal, purchases journal, cash receipts journal, cash disbursements journal, and general journal. On June 5, Houst purchased merchandise priced at...

Study smarter with the SolutionInn App


