Instructions: Present the solutions, with appropriate supporting calculations, for each of the following independent problems. A....
Fantastic news! We've Found the answer you've been seeking!
Question:
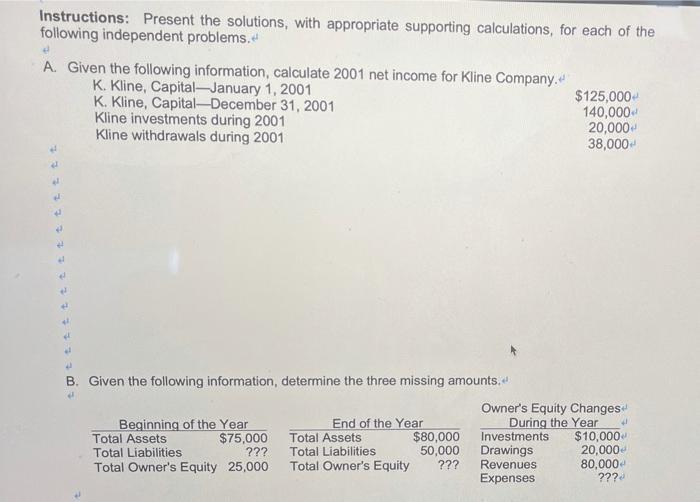
Transcribed Image Text:
Instructions: Present the solutions, with appropriate supporting calculations, for each of the following independent problems. A. Given the following information, calculate 2001 net income for Kline Company. K. Kline, Capital-January 1, 2001 K. Kline, Capital-December 31, 2001 Kline investments during 2001 Kline withdrawals during 2001 B. Given the following information, determine the three missing amounts. d Beginning of the Year Total Assets $75,000 Total Liabilities ??? Total Owner's Equity 25,000 End of the Year Total Assets Total Liabilities Total Owner's Equity $80,000 50,000 ??? $125,000+ 140,000 20,000+ 38,000 Owner's Equity Changes During the Year Investments $10,000 20,000 80,000 ??? Drawings Revenues Expenses Instructions: Present the solutions, with appropriate supporting calculations, for each of the following independent problems. A. Given the following information, calculate 2001 net income for Kline Company. K. Kline, Capital-January 1, 2001 K. Kline, Capital-December 31, 2001 Kline investments during 2001 Kline withdrawals during 2001 B. Given the following information, determine the three missing amounts. d Beginning of the Year Total Assets $75,000 Total Liabilities ??? Total Owner's Equity 25,000 End of the Year Total Assets Total Liabilities Total Owner's Equity $80,000 50,000 ??? $125,000+ 140,000 20,000+ 38,000 Owner's Equity Changes During the Year Investments $10,000 20,000 80,000 ??? Drawings Revenues Expenses
Expert Answer:
Answer rating: 100% (QA)
Question Note Question is to be solved using State of Changes in Owners Equity A Calculation of net ... View the full answer

Related Book For 

Project Management A Managerial Approach
ISBN: 978-0470533024
8th edition
Authors: Jack R. Meredith, Samuel J. Mantel Jr.
Posted Date:
Students also viewed these finance questions
-
Calculate Tucker Company's current ratio, given the following information about its assets and liabilities (round to two decimal places) and compare it to the industry average current ratio of 2.2....
-
Given the following information for Smashville, Inc., construct an income statement for the year: Cost of goods sold: .......... $149,000 Investment income: .......... $1,900 Net sales:...
-
A teller at a drive-up window at a bank had the following service times (in minutes) for 20 randomly selected customers: What are the 3-sigma control limits? Select one: a. LCL = 4.385, UCL = 4.615...
-
A vertical well is drilled through a stratigraphic section twice (repeated section). What type of fault can we infer, and why can we not explain this by folding?
-
Central Community College is preparing a brochure to persuade prospective students to consider taking classes. The college doesnt have the money for full-scale document testing. What free or...
-
Bernard Creighton is the controller for Creighton Hardware Store. In putting together the cash budget for the fourth quarter of the year, he has assembled the following data: a. Sales July (actual)...
-
How might a hacker access and manipulate a digital device for illegal purposes? Are the Internet of Things (IoT) devices at risk for hacker access and manipulation?
-
Comparative financial statement data for Carmono Company follow: For 2009, the company reported net income as follows: Dividends of $14 were declared and paid during 2009. Required: 1. Using the...
-
We need to map following EERD into RELATIONAL MODEL. Map the following EERD into relational model ArtistNumber Artist Name N FormationDate Group Group-Member SoloPerformer BirthDate JoinedDate...
-
Consider the following linear programming problem Max 8X + 7Y s.t. 15X + 5Y < 75 10X + 6Y < 60 X + Y < 8 X, Y 0 a. Use a graph to show each constraint and the feasible region. b. Identify the...
-
Using the annexes provided (specially #5), compare financial ratios over the years. Prepare an analysis for each ratio working for the years 2014 to 2017. a. How do the liquidity ratios compare for...
-
14. As implied by the speaker, those possessing "great wealth" fear change because reforms may Question 14 options: a. create a more equitable society b. restrict profits to only an enterprising few...
-
Solve the problem and show the complete solution. A triangle has sides 23 and 15 ft with an included angle of 30. If the linear measurements may be in error by as much as one inch and the angle by as...
-
An intersection has two lanes on each approach of the major street and two lanes on each approach of the minor street. The major road speed limit is 55 mph. Summary traffic volume data is provided in...
-
The death penalty should be legalized in Canada: CON Title of your debate and which side of the argument you are taking Explanation of what you are planning to argue. Provide examples of how you...
-
Describe the American judicial system (civil criminal and juvenile) their jurisdiction, development and structure Analyze the function and dynamics of the courtroom work group Identify judicial...
-
As an educator you will be part of the whole service team, the educator team and a room team. Think about responsibilities as a member of the whole team. Provide one example of key...
-
As water moves through the hydrologic cycle, water quality changes are common because of natural phenomena or anthropogenic pollution. Using Figure 11.1, describe how water-quality changes occur...
-
What do you think are the reasons BP took such a relaxed attitude toward the Gulf well compared to NASA?
-
What might be the weaknesses of this new approach?
-
Was the life cycle for this project S-shaped, J-shaped, or something else? Considering just the purpose of the river dredging as a project itself, what was the shape of its life cycle?
-
Four moles of nitrogen and one mole of oxygen at \(P=1 \mathrm{~atm}\) and \(T=300 \mathrm{~K}\) are mixed together to form air at the same pressure and temperature. Calculate the entropy of mixing...
-
A mole of argon and a mole of helium are contained in vessels of equal volume. If argon is at \(300 \mathrm{~K}\), what should the temperature of helium be so that the two have the same entropy?
-
If the two gases considered in the mixing process of Section 1.5 were initially at different temperatures, say \(T_{1}\) and \(T_{2}\), what would the entropy of mixing be in that case? Would the...

Study smarter with the SolutionInn App


