Gold has always been a highly prized metal, widely used from the beginning of history as...
Fantastic news! We've Found the answer you've been seeking!
Question:
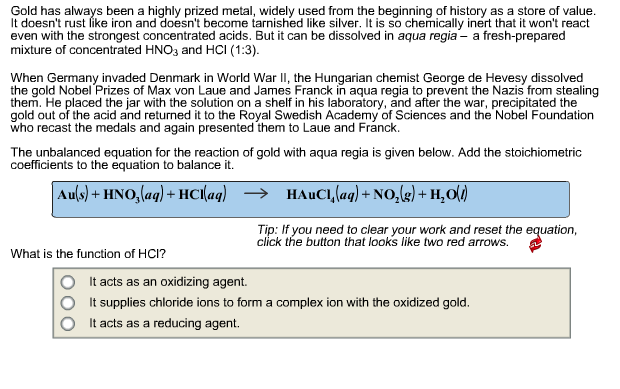
Transcribed Image Text:
Gold has always been a highly prized metal, widely used from the beginning of history as a store of value. It doesn't rust like iron and doesn't become tarnished like silver. It is so chemically inert that it won't react even with the strongest concentrated acids. But it can be dissolved in aqua regia- a fresh-prepared mixture of concentrated HNO3 and HCI (1:3). When Germany invaded Denmark in World War II, the Hungarian chemist George de Hevesy dissolved the gold Nobel Prizes of Max von Laue and James Franck in aqua regia to prevent the Nazis from stealing them. He placed the jar with the solution on a shelf in his laboratory, and after the war, precipitated the gold out of the acid and returned it to the Royal Swedish Academy of Sciences and the Nobel Foundation who recast the medals and again presented them to Laue and Franck. The unbalanced equation for the reaction of gold with aqua regia is given below. Add the stoichiometric coefficients to the equation to balance it. Au(s) + HNO3(aq) + HCl(aq) What is the function of HCI? HAUCI, (aq) + NO₂(g) + H₂O(l) Tip: If you need to clear your work and reset the equation, click the button that looks like two red arrows. It acts as an oxidizing agent. It supplies chloride ions to form a complex ion with the oxidized gold. It acts as a reducing agent. Gold has always been a highly prized metal, widely used from the beginning of history as a store of value. It doesn't rust like iron and doesn't become tarnished like silver. It is so chemically inert that it won't react even with the strongest concentrated acids. But it can be dissolved in aqua regia- a fresh-prepared mixture of concentrated HNO3 and HCI (1:3). When Germany invaded Denmark in World War II, the Hungarian chemist George de Hevesy dissolved the gold Nobel Prizes of Max von Laue and James Franck in aqua regia to prevent the Nazis from stealing them. He placed the jar with the solution on a shelf in his laboratory, and after the war, precipitated the gold out of the acid and returned it to the Royal Swedish Academy of Sciences and the Nobel Foundation who recast the medals and again presented them to Laue and Franck. The unbalanced equation for the reaction of gold with aqua regia is given below. Add the stoichiometric coefficients to the equation to balance it. Au(s) + HNO3(aq) + HCl(aq) What is the function of HCI? HAUCI, (aq) + NO₂(g) + H₂O(l) Tip: If you need to clear your work and reset the equation, click the button that looks like two red arrows. It acts as an oxidizing agent. It supplies chloride ions to form a complex ion with the oxidized gold. It acts as a reducing agent.
Expert Answer:
Related Book For 

Valuation The Art and Science of Corporate Investment Decisions
ISBN: 978-0133479522
3rd edition
Authors: Sheridan Titman, John D. Martin
Posted Date:
Students also viewed these chemistry questions
-
The change in retained earnings from the beginning of the year to the end of the year is the result of net earnings minus dividends for the year. Calculate the missing amounts in the reconciliation...
-
The Bureau of Finance has always been a relaxed place to work. Employees are allowed 60 minutes for a lunch break and are not required to sign or punch in or out. Recently the head of the Bureau has...
-
The change in retained earnings from the beginning of the year to the end of the year is the result of net earnings minus dividends for the year. These changes are part of the information presented...
-
What is the relationship between group norms and group cohesiveness? What roles do both cohesiveness and norms plan in shaping group performance?
-
The 2001 annual report for General Motors Corporation contained the following note: Note 3. Significant Accounting Policies Property, Net Property, plant, and equipment, including internal use...
-
D. Lawrance designs and manufactures fashionable mens clothing. For the coming year, the company has scheduled production of 40,000 suede jackets. Budgeted costs for this product are as follows: The...
-
What are the different types of consulting and litigation support activities for fraud and forensic accounting professionals?
-
As a recently hired accountant for a small business, Bearing, Inc., you are provided with last year's balance sheet, income statement, and post-closing trial balance to familiarize yourself with the...
-
A small business has a cost function C (z) = 722-217 +2,352 and a revenue function of R(x) 6. At the break-even points the values of in increasing order (smallest one first) are Number and Number
-
The TimpRiders LP has operated a motorcycle dealership for a number of years. Lance is the limited partner, Francesca is the general partner, and they share capital and profits equally. Francesca...
-
The following financial information has been provided for Up & Down Ltd. for its year ended September 30, 2020. Statement of Income Revenue Depreciation/Amortization Administration and other expenses...
-
A home buyer will take out a mortgage of $280,000 for a new house. The mortgage will feature 30 years of monthly payments. The mortgage carries a 4.80% APR (with monthly compounding). What will be...
-
A student invests $1,000 today in a savings account that pays 2.4% APR with monthly compounding. The student will leave the money in the account for exactly 10 years. What will be the balance at the...
-
A steel ball of mass 0.790 kg is fastened to a cord that is 90.0 cm long and fixed at the far end. The ball is then released when the cord is horizontal, as shown in the figure. At the bottom of its...
-
You have just received notification that you have won the $2.04 million first prize in the Centennial Lottery. However, the prize will be awarded on your 100th birthday (assuming you're around to...
-
Imprudential, Inc., has an unfunded pension liability of $773 million that must be paid in 10 years. To assess the value of the firm's stock, financial analysts want to discount this liability back...
-
The risk-free interest rate is the same as the data in Problem 4.5. What is the value of the FRA that begins after a year when it pays LIBOR for three months on a principal of 1,000,000 months of...
-
The figure shows a bolted lap joint that uses SAE grade 8 bolts. The members are made of cold-drawn AISI 1040 steel. Find the safe tensile shear load F that can be applied to this connection if the...
-
Google Inc. of Mountain View, California, operates the most popular and power-ful search engine on the Web. The company went public using an unconventional Dutch auction method on August 19, 2004....
-
In 2006, the major airline carriers, with the principal exception of Southwest Airlines (LUV), continued to be in dire financial condition following the attack on the World Trade Center in 2001. a....
-
The destruction that Hurricane Katrina brought to the Gulf Coast in 2005 devastated the city of New Orleans as well as the Mississippi Gulf Coast. The burgeoning casino gambling industry along the...
-
Obtain a copy of the ACAS Code of Practice 1: Disciplinary and Grievance Procedures (2009). Use this, and our suggestions in this chapter, to rewrite or modify either of the procedures you used for...
-
One of the disadvantages of the IEEET1 exciter is following a fault the terminal voltage does not necessarily return to its prefault value. Using PowerWorld Simulator case Problem 12_3 determine the...
-
Rework Example 13.6 if the source impedance at the sending end of line \(\mathrm{A}\) is \(\mathrm{Z}_{\mathrm{G}}=\mathrm{Z}_{\mathrm{A}} / 4=100 \Omega\), and the receiving end of line...

Study smarter with the SolutionInn App



