How many milliliters of water at 23 C with a density of 1.00 g/mL must be...
Fantastic news! We've Found the answer you've been seeking!
Question:
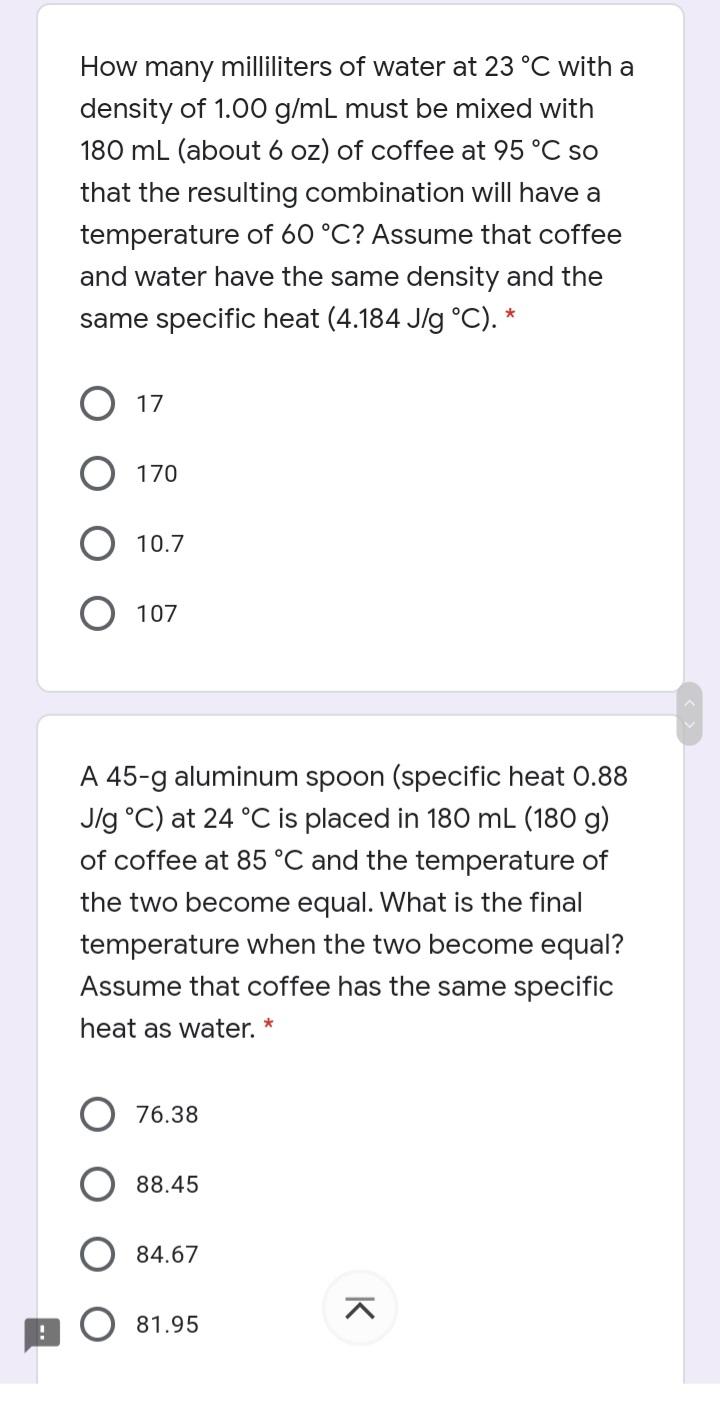
Transcribed Image Text:
How many milliliters of water at 23 °C with a density of 1.00 g/mL must be mixed with 180 mL (about 6 oz) of coffee at 95 °C so that the resulting combination will have a temperature of 60 °C? Assume that coffee and water have the same density and the same specific heat (4.184 J/g °C). * O 17 170 10.7 107 A 45-g aluminum spoon (specific heat 0.88 Jlg °C) at 24 °C is placed in 180 mL (180 g) of coffee at 85 °C and the temperature of the two become equal. What is the final temperature when the two become equal? Assume that coffee has the same specific heat as water. * 76.38 88.45 84.67 81.95 K How many milliliters of water at 23 °C with a density of 1.00 g/mL must be mixed with 180 mL (about 6 oz) of coffee at 95 °C so that the resulting combination will have a temperature of 60 °C? Assume that coffee and water have the same density and the same specific heat (4.184 J/g °C). * O 17 170 10.7 107 A 45-g aluminum spoon (specific heat 0.88 Jlg °C) at 24 °C is placed in 180 mL (180 g) of coffee at 85 °C and the temperature of the two become equal. What is the final temperature when the two become equal? Assume that coffee has the same specific heat as water. * 76.38 88.45 84.67 81.95 K
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these physics questions
-
How many milliliters of 0.163 M NaCl are required to give 0.0958 g of sodium chloride?
-
How many milliliters of 0.150 M H2SO4 (sulfuric acid) are required to react with 8.20 g of sodium hydrogen carbonate, NaHCO3, according to the following equation? H2SO4(aq) + 2NaHCO3(aq) Na2SO4(aq)...
-
How many milliliters of 0.250 M KMnO4 are needed to react with 3.36 g of iron(II) sulfate, FeSO4? The reaction is as follows: 10FeSO4(aq) + 2KMnO4(aq) + 8H2SO4(aq) 5Fe2(SO4)3(aq) + 2MnSO4(aq) +...
-
Timco is considering the construction of a new retail outlet. The construction cost will be 400000. Net working capital will increase by 10000. The depreciation is 10 year MACRS. The new location...
-
Compute the maturity value of a 150-day, 6% promissory note with a face value of $5000 dated August 5.
-
Why do certain seed investors prefer convertible notes?
-
What parameter in a vibration spectrum is most important for separating individual fault frequencies?
-
In one stage of an annealing process, 304 stainless steel sheet is taken from 300 K to 1250 K as it passes through an electrically heated oven at a speed of V, = 10 mm/s. The sheet thickness and...
-
Define the Role of the Modern Forensic Psychologist ? What are the basic Competencies and Training Models in Forensic Psychology ?
-
Bronys Bikes maintains two general demand deposit bank accounts and an imprest payroll account. One of the general bank accounts and the payroll account are with Dallas Dollar Bank. The second demand...
-
A heat exchanger is used to transfer heat from a hot fluid to a cold fluid. The hot fluid has a flow rate of 5 kg/s and a temperature of 200C, while the cold fluid has a flow rate of 10 kg/s and a...
-
To obtain new computers, Liberia accepts bids from U.S. firms, including Macro Corporation and Micro, Inc. Macro wins the contract. Alleging impropriety, Micro files a suit in a U.S. court against...
-
A simple random sample of size \(n=25\) is drawn from a population that is normally distributed. The sample variance is found to be \(s^{2}=3.97\). Construct a \(95 \%\) confidence interval for the...
-
Figure 1.5 shows a micrograph of chromosomes from a normal human cell. If you created this kind of image using a cell from a person with Down syndrome, what would you expect to see? Figure 1.5 : 1 2...
-
A patient is prescribed a brand name drug. The patient asks the pharmacist if generics are available. The pharmacists research shows generics are available but unapproved by the FDA. The pharmacist...
-
What was the change in Global Conglomerates book value of equity from 2021 to 2022 according to Table 2.1? Does this imply that the market price of Globals shares increased in 2022? Explain. TABLE...
-
The Great Eastern Shoe Company produces athletic shoes for a variety of companies. Currently, they are reexamining all of their factories' efficiencies. They are doing this on a country-by-country...
-
XYZ Inc. a calendar year, accrual basis corporation, had the following items during 2021: Gross revenue from operations Cost of goods sold $420,000 ($180,000) $9,000 LT capital gain .LT capital...
-
Obtain the pH corresponding to the following hydronium-ion concentrations. a. 1.0 108 M b. 5.0 1012 M c. 7.5 103 M d. 6.35 109 M
-
Calculate the equilibrium constant K for the following reaction at 25C from standard electrode potentials. Sn4+(aq) + 2Hg(l Sn2+(aq) + Hg2 2+(aq) The equation is not balanced.
-
A 75.0-g sample of a pure liquid, liquid A, with a density of 3.00 g/mL is mixed with a 50.0-mL sample of a pure liquid, liquid B, with a density of 2.00 g/mL. What is the total volume of the...
-
Find the heat transfer rate \(\mathrm{q}_{\mathrm{w}}\) at \(\mathrm{x}=10 \mathrm{~cm}\) and \(100 \mathrm{~cm}\) for the flat plate given in Problem 7.31. Problem 7.31 A flat plate of \(4...
-
Obtain the lift and propulsive force coefficients of an airfoil given in Example 8.6, and compare the results with Problem 8.30. Assume the profile pitches about midchord. Example 8.6 The NACA 0012...
-
What are the values of the feathering parameters for the airfoils given by Examples 8.5 and 8.6? Examples 8.5 Assume an airfoil pitching about its leading edge and plunging with \(k=0.35\) as follows...

Study smarter with the SolutionInn App


