Human serum albumin (HSA) has a molecular weight of 66,000 g/mol. One compartment contains a solution...
Fantastic news! We've Found the answer you've been seeking!
Question:
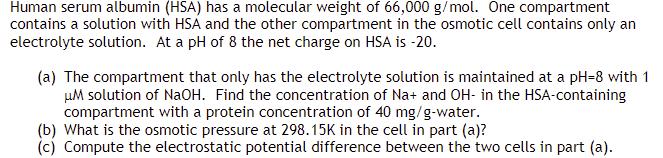
Transcribed Image Text:
Human serum albumin (HSA) has a molecular weight of 66,000 g/mol. One compartment contains a solution with HSA and the other compartment in the osmotic cell contains only an electrolyte solution. At a pH of 8 the net charge on HSA is -20. (a) The compartment that only has the electrolyte solution is maintained at a pH-8 with 1 μM solution of NaOH. Find the concentration of Na+ and OH- in the HSA-containing compartment with a protein concentration of 40 mg/g-water. (b) What is the osmotic pressure at 298.15K in the cell in part (a)? (c) Compute the electrostatic potential difference between the two cells in part (a). Human serum albumin (HSA) has a molecular weight of 66,000 g/mol. One compartment contains a solution with HSA and the other compartment in the osmotic cell contains only an electrolyte solution. At a pH of 8 the net charge on HSA is -20. (a) The compartment that only has the electrolyte solution is maintained at a pH-8 with 1 μM solution of NaOH. Find the concentration of Na+ and OH- in the HSA-containing compartment with a protein concentration of 40 mg/g-water. (b) What is the osmotic pressure at 298.15K in the cell in part (a)? (c) Compute the electrostatic potential difference between the two cells in part (a).
Expert Answer:
Answer rating: 100% (QA)
Answer Lets solve each part step by step a Concentration of Na and OH in the HSAcontaining compartme... View the full answer

Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Posted Date:
Students also viewed these chemical engineering questions
-
Various amino acids have utility as food additives and in medical applications. They are often synthesized by fermentation using a specific microorganism to convert a substrate (e.g., a sugar) into...
-
A certain compound has a molecular weight of 83 and contains nitrogen. Its infrared spectrum contains a moderately strong peak at 2270 cm-1. Its 1H and 13C NMR spectra are shown in Figure 20.10. What...
-
Michael Jackson was hired in January as the new Operations Manager for Just Collect It, Inc, a Texas toy assembly plant also known as JCI. As the Operations Manager Michaels duties included contract...
-
Graph each list of numbers on the given number line. See Example 1. 1. 2. -1, .3 + + + 4 2 -5 -4 -3 -2 -1 0 1 + en 3 10 -5 -4 -3 -2 -1 en 3,
-
In Exercises, a probability density function of a random variable is defined. Find the expected value, the variance, and the standard deviation. Round answers to the nearest hundredth. (x) = 4x -5 ;...
-
(a) Does it make sense to talk about a dielectric constant for a conductor? If so, what value does the constant have? (b) What is the breakdown threshold for a conductor?
-
During 2012, Maverick Inc. became involved in a tax dispute with the IRS. Mavericks attorneys have indicated that they believe it is probable that Maverick will lose this dispute. They also believe...
-
1. Define ADT (Abstract Data Type)? 2. Mention the features of ADT.? 3. Define List ADT? 4. What are the ways of implementing linked list? 5. What are the types of linked lists?
-
Marider Industries makes two types of windbreaker jackets: one for spring and one for winter. Marider uses a traditional costing system and is considering switching to an Activity-Based Costing...
-
A commercial bank has provided the balance sheet below. It has no off-balance sheet activities. Commercial loans have a 100% loan-to-value risk weight and construction loans have a 50% loan-to value...
-
Is being a civil engineer worth it? Explain why?
-
Data for Hermann Corporation are shown below: Selling price Variable expenses Contribution margin Percent Per Unit of Sales $ 60 39 100% 65 $ 21 35% Fixed expenses are $72,000 per month and the...
-
The following Trial Balance was extracted from the books of Hope Enterprise as at 31 December 2022. Sales Purchases Inventory as at 1 January 2022 Motor vehicles Office equipment Accounts receivable...
-
ps://mybscbryantstratton.edu/webapps/assessment/take/launch.jsp?course_assessment_id=_361377_18course_id=_93... A Question Completion Status: QUESTION 10 8.5 points Compose a statement that correctly...
-
2. Enlist the assistance of at least four volunteers to undertake the role of the members of Team A and Team B. Each team must have at least two members. Discuss with your volunteers the roles they...
-
1. What factors play a role in post-traumatic stress disorder? (select all that are correct, 3 correct) A. Those with PTSD often have chronic tension, anxiety, and health problems. B.Th. memory of a...
-
From a medical tourist perspective, compare Shouldice with the traditional hospital in terms of the key factors of competition. Using Table 15-3, why would Shouldice attract patients from outside the...
-
Which is the greater mass, 3257 mg or 0.000475 kg? Explain.
-
(A) Creatinine is a by-product of nitrogen metabolism and can be used to provide an indication of renal function. A 4.04 g sample of creatinine is dissolved in enough water to make 100.0 mL of...
-
A laboratory high vacuum system is capable of evacuating a vessel to the point that the amount of gas remaining is 5.0 x 10 9 molecules per cubic meter. What is the residual pressure in pascals?
-
Financial balances for the car hire business of Terry's Wedding Cars on 31 March 2024 are provided below in a table in accounting equation form similar to the chapter illustrations. During April, the...
-
Trans Clothing Alterations began operations on 1 August 2024 and completed the following transactions during the first month. 1. Tran deposited \($18\) 000 of her personal funds in a current account...
-
Finesse Fitness was established on 1 April 2024 with an initial investment of $60000 by the owner, Daniel Hewitt. During the first few months of business, the owner employed a student studying...

Study smarter with the SolutionInn App


