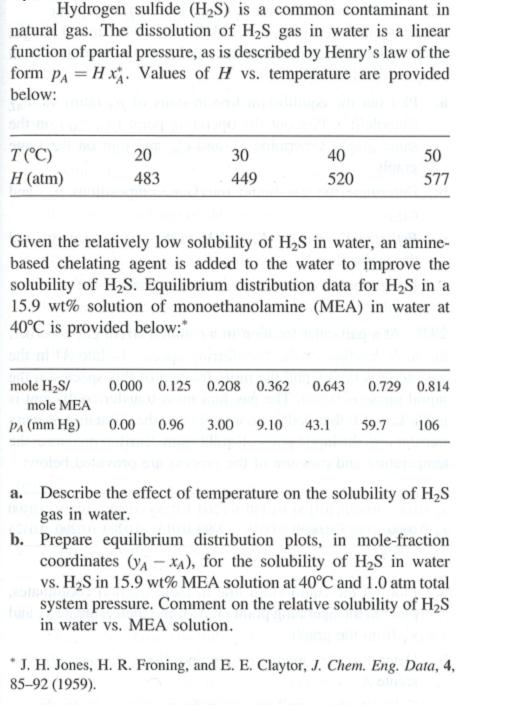
Hydrogen sulfide (HS) is a common contaminant in natural gas. The dissolution of HS gas in...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Hydrogen sulfide (H₂S) is a common contaminant in natural gas. The dissolution of H₂S gas in water is a linear function of partial pressure, as is described by Henry's law of the form PAHx. Values of H vs. temperature are provided below: T (°C) H (atm) 20 483 30 449 mole H₂S/ 40 520 50 577 Given the relatively low solubility of H₂S in water, an amine- based chelating agent is added to the water to improve the solubility of H₂S. Equilibrium distribution data for H₂S in a 15.9 wt% solution of monoethanolamine (MEA) in water at 40°C is provided below:* 0.000 0.125 0.208 0.362 0.643 0.729 0.814 mole MEA PA (mm Hg) 0.00 0.96 3.00 9.10 43.1 59.7 106 a. Describe the effect of temperature on the solubility of H₂S gas in water. b. Prepare equilibrium distribution plots, in mole-fraction coordinates (VA-XA), for the solubility of H₂S in water vs. H₂S in 15.9 wt% MEA solution at 40°C and 1.0 atm total system pressure. Comment on the relative solubility of H₂S in water vs. MEA solution. J. H. Jones, H. R. Froning, and E. E. Claytor, J. Chem. Eng. Data, 4, 85-92 (1959). Hydrogen sulfide (H₂S) is a common contaminant in natural gas. The dissolution of H₂S gas in water is a linear function of partial pressure, as is described by Henry's law of the form PAHx. Values of H vs. temperature are provided below: T (°C) H (atm) 20 483 30 449 mole H₂S/ 40 520 50 577 Given the relatively low solubility of H₂S in water, an amine- based chelating agent is added to the water to improve the solubility of H₂S. Equilibrium distribution data for H₂S in a 15.9 wt% solution of monoethanolamine (MEA) in water at 40°C is provided below:* 0.000 0.125 0.208 0.362 0.643 0.729 0.814 mole MEA PA (mm Hg) 0.00 0.96 3.00 9.10 43.1 59.7 106 a. Describe the effect of temperature on the solubility of H₂S gas in water. b. Prepare equilibrium distribution plots, in mole-fraction coordinates (VA-XA), for the solubility of H₂S in water vs. H₂S in 15.9 wt% MEA solution at 40°C and 1.0 atm total system pressure. Comment on the relative solubility of H₂S in water vs. MEA solution. J. H. Jones, H. R. Froning, and E. E. Claytor, J. Chem. Eng. Data, 4, 85-92 (1959). Hydrogen sulfide (H₂S) is a common contaminant in natural gas. The dissolution of H₂S gas in water is a linear function of partial pressure, as is described by Henry's law of the form PAHx. Values of H vs. temperature are provided below: T (°C) H (atm) 20 483 30 449 mole H₂S/ 40 520 50 577 Given the relatively low solubility of H₂S in water, an amine- based chelating agent is added to the water to improve the solubility of H₂S. Equilibrium distribution data for H₂S in a 15.9 wt% solution of monoethanolamine (MEA) in water at 40°C is provided below:* 0.000 0.125 0.208 0.362 0.643 0.729 0.814 mole MEA PA (mm Hg) 0.00 0.96 3.00 9.10 43.1 59.7 106 a. Describe the effect of temperature on the solubility of H₂S gas in water. b. Prepare equilibrium distribution plots, in mole-fraction coordinates (VA-XA), for the solubility of H₂S in water vs. H₂S in 15.9 wt% MEA solution at 40°C and 1.0 atm total system pressure. Comment on the relative solubility of H₂S in water vs. MEA solution. J. H. Jones, H. R. Froning, and E. E. Claytor, J. Chem. Eng. Data, 4, 85-92 (1959). Hydrogen sulfide (H₂S) is a common contaminant in natural gas. The dissolution of H₂S gas in water is a linear function of partial pressure, as is described by Henry's law of the form PAHx. Values of H vs. temperature are provided below: T (°C) H (atm) 20 483 30 449 mole H₂S/ 40 520 50 577 Given the relatively low solubility of H₂S in water, an amine- based chelating agent is added to the water to improve the solubility of H₂S. Equilibrium distribution data for H₂S in a 15.9 wt% solution of monoethanolamine (MEA) in water at 40°C is provided below:* 0.000 0.125 0.208 0.362 0.643 0.729 0.814 mole MEA PA (mm Hg) 0.00 0.96 3.00 9.10 43.1 59.7 106 a. Describe the effect of temperature on the solubility of H₂S gas in water. b. Prepare equilibrium distribution plots, in mole-fraction coordinates (VA-XA), for the solubility of H₂S in water vs. H₂S in 15.9 wt% MEA solution at 40°C and 1.0 atm total system pressure. Comment on the relative solubility of H₂S in water vs. MEA solution. J. H. Jones, H. R. Froning, and E. E. Claytor, J. Chem. Eng. Data, 4, 85-92 (1959). Hydrogen sulfide (H₂S) is a common contaminant in natural gas. The dissolution of H₂S gas in water is a linear function of partial pressure, as is described by Henry's law of the form PAHx. Values of H vs. temperature are provided below: T (°C) H (atm) 20 483 30 449 mole H₂S/ 40 520 50 577 Given the relatively low solubility of H₂S in water, an amine- based chelating agent is added to the water to improve the solubility of H₂S. Equilibrium distribution data for H₂S in a 15.9 wt% solution of monoethanolamine (MEA) in water at 40°C is provided below:* 0.000 0.125 0.208 0.362 0.643 0.729 0.814 mole MEA PA (mm Hg) 0.00 0.96 3.00 9.10 43.1 59.7 106 a. Describe the effect of temperature on the solubility of H₂S gas in water. b. Prepare equilibrium distribution plots, in mole-fraction coordinates (VA-XA), for the solubility of H₂S in water vs. H₂S in 15.9 wt% MEA solution at 40°C and 1.0 atm total system pressure. Comment on the relative solubility of H₂S in water vs. MEA solution. J. H. Jones, H. R. Froning, and E. E. Claytor, J. Chem. Eng. Data, 4, 85-92 (1959). Hydrogen sulfide (H₂S) is a common contaminant in natural gas. The dissolution of H₂S gas in water is a linear function of partial pressure, as is described by Henry's law of the form PAHx. Values of H vs. temperature are provided below: T (°C) H (atm) 20 483 30 449 mole H₂S/ 40 520 50 577 Given the relatively low solubility of H₂S in water, an amine- based chelating agent is added to the water to improve the solubility of H₂S. Equilibrium distribution data for H₂S in a 15.9 wt% solution of monoethanolamine (MEA) in water at 40°C is provided below:* 0.000 0.125 0.208 0.362 0.643 0.729 0.814 mole MEA PA (mm Hg) 0.00 0.96 3.00 9.10 43.1 59.7 106 a. Describe the effect of temperature on the solubility of H₂S gas in water. b. Prepare equilibrium distribution plots, in mole-fraction coordinates (VA-XA), for the solubility of H₂S in water vs. H₂S in 15.9 wt% MEA solution at 40°C and 1.0 atm total system pressure. Comment on the relative solubility of H₂S in water vs. MEA solution. J. H. Jones, H. R. Froning, and E. E. Claytor, J. Chem. Eng. Data, 4, 85-92 (1959). Hydrogen sulfide (H₂S) is a common contaminant in natural gas. The dissolution of H₂S gas in water is a linear function of partial pressure, as is described by Henry's law of the form PAHx. Values of H vs. temperature are provided below: T (°C) H (atm) 20 483 30 449 mole H₂S/ 40 520 50 577 Given the relatively low solubility of H₂S in water, an amine- based chelating agent is added to the water to improve the solubility of H₂S. Equilibrium distribution data for H₂S in a 15.9 wt% solution of monoethanolamine (MEA) in water at 40°C is provided below:* 0.000 0.125 0.208 0.362 0.643 0.729 0.814 mole MEA PA (mm Hg) 0.00 0.96 3.00 9.10 43.1 59.7 106 a. Describe the effect of temperature on the solubility of H₂S gas in water. b. Prepare equilibrium distribution plots, in mole-fraction coordinates (VA-XA), for the solubility of H₂S in water vs. H₂S in 15.9 wt% MEA solution at 40°C and 1.0 atm total system pressure. Comment on the relative solubility of H₂S in water vs. MEA solution. J. H. Jones, H. R. Froning, and E. E. Claytor, J. Chem. Eng. Data, 4, 85-92 (1959). Hydrogen sulfide (H₂S) is a common contaminant in natural gas. The dissolution of H₂S gas in water is a linear function of partial pressure, as is described by Henry's law of the form PAHx. Values of H vs. temperature are provided below: T (°C) H (atm) 20 483 30 449 mole H₂S/ 40 520 50 577 Given the relatively low solubility of H₂S in water, an amine- based chelating agent is added to the water to improve the solubility of H₂S. Equilibrium distribution data for H₂S in a 15.9 wt% solution of monoethanolamine (MEA) in water at 40°C is provided below:* 0.000 0.125 0.208 0.362 0.643 0.729 0.814 mole MEA PA (mm Hg) 0.00 0.96 3.00 9.10 43.1 59.7 106 a. Describe the effect of temperature on the solubility of H₂S gas in water. b. Prepare equilibrium distribution plots, in mole-fraction coordinates (VA-XA), for the solubility of H₂S in water vs. H₂S in 15.9 wt% MEA solution at 40°C and 1.0 atm total system pressure. Comment on the relative solubility of H₂S in water vs. MEA solution. J. H. Jones, H. R. Froning, and E. E. Claytor, J. Chem. Eng. Data, 4, 85-92 (1959). Hydrogen sulfide (H₂S) is a common contaminant in natural gas. The dissolution of H₂S gas in water is a linear function of partial pressure, as is described by Henry's law of the form PAHx. Values of H vs. temperature are provided below: T (°C) H (atm) 20 483 30 449 mole H₂S/ 40 520 50 577 Given the relatively low solubility of H₂S in water, an amine- based chelating agent is added to the water to improve the solubility of H₂S. Equilibrium distribution data for H₂S in a 15.9 wt% solution of monoethanolamine (MEA) in water at 40°C is provided below:* 0.000 0.125 0.208 0.362 0.643 0.729 0.814 mole MEA PA (mm Hg) 0.00 0.96 3.00 9.10 43.1 59.7 106 a. Describe the effect of temperature on the solubility of H₂S gas in water. b. Prepare equilibrium distribution plots, in mole-fraction coordinates (VA-XA), for the solubility of H₂S in water vs. H₂S in 15.9 wt% MEA solution at 40°C and 1.0 atm total system pressure. Comment on the relative solubility of H₂S in water vs. MEA solution. J. H. Jones, H. R. Froning, and E. E. Claytor, J. Chem. Eng. Data, 4, 85-92 (1959). Hydrogen sulfide (H₂S) is a common contaminant in natural gas. The dissolution of H₂S gas in water is a linear function of partial pressure, as is described by Henry's law of the form PAHx. Values of H vs. temperature are provided below: T (°C) H (atm) 20 483 30 449 mole H₂S/ 40 520 50 577 Given the relatively low solubility of H₂S in water, an amine- based chelating agent is added to the water to improve the solubility of H₂S. Equilibrium distribution data for H₂S in a 15.9 wt% solution of monoethanolamine (MEA) in water at 40°C is provided below:* 0.000 0.125 0.208 0.362 0.643 0.729 0.814 mole MEA PA (mm Hg) 0.00 0.96 3.00 9.10 43.1 59.7 106 a. Describe the effect of temperature on the solubility of H₂S gas in water. b. Prepare equilibrium distribution plots, in mole-fraction coordinates (VA-XA), for the solubility of H₂S in water vs. H₂S in 15.9 wt% MEA solution at 40°C and 1.0 atm total system pressure. Comment on the relative solubility of H₂S in water vs. MEA solution. J. H. Jones, H. R. Froning, and E. E. Claytor, J. Chem. Eng. Data, 4, 85-92 (1959).
Expert Answer:

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemical engineering questions
-
The table below presents equilibrium distribution data for four gaseous solutes dissolved in water, using air as the carrier gas: a. Using a spreadsheet to perform the calculations, prepare a graph...
-
Natural gas frequently contains hydrogen sulfide, H2S. H2S is removed from natural gas by passing it through aqueous ethanolamine, HOCH2CH2NH2 (an ammonia derivative), which reacts with the hydrogen...
-
Hydrogen sulfide can be removed from natural gas by the reaction 2H2S(g) + SO2(g) 3S(s) + 2H2O(g) Calculate Go and K (at 298 K) for this reaction. Would this reaction be favored at a high or low...
-
Exercises 5-8: Sort the list of numbers from smallest to largest and display the result in a table. (a) Determine the maximum and minimum values. (b) Calculate the mean and median. Round each result...
-
Half a century ago men were far more likely to be the breadwinner and women were far more likely to be homemakers. Has this changed or are men still more likely than women to work full time outside...
-
Implement the unify algorithm of Section 2.3.2 in the computer language of your choice. Data from section 2.3.2 To apply inference rules such as modus ponens, an inference system must be able to...
-
The salaries (in thousand dollars) of a sample of 10 employees Organize the data using the indicated type of graph. Describe any patterns. 225 410 368 310 228 298 361 159 486 296
-
Cost classification and target profit Walts Woodwork Company makes and sells wooden shelves. Walts carpenters make the shelves in the companys rented building. Walt has a separate office at another...
-
A car is traveling around a banked curve with a radius of 100 meters and a banking angle of 30 degrees at a speed of 40 m/s. What is the coefficient of friction between the tires and the road that...
-
Manager's Hot Seat Negotiation: Thawing the Salary Freeze from the video create a report. What could Katherine have done or said differently?
-
You are to enter the following transactions in the books of PP Enterprise, post to accounts, balance off the accounts, extract a trial balance and prepare profit or loss statement and balance sheet...
-
Explain why a little inflation is always necessary to grease the wheels of the economy.
-
Imagine a PPF for two goods: guns and butter. A natural disaster hits and reduces our ability to produce both weapons and butter. What will be the effect?
-
How did Western domination affect South Asia, the Islamic heartland, and Africa in the 19th century? Explain
-
Instructions: Revise the message below so that it has improved tone and formality. Also revise it for better organization (use the direct approach). Post your revision to OneNote. From: H & M Calgary...
-
Suppose that if your income increases by $100, all households in Normalia increase their expenses by $50. Instructions: Round your answers to one decimal place. The MPC in Normalia is: MPS in...
-
Aim is to transmit water from the source to the end point. Option is to use gravity pipeline system (may use tools i.e. pump, air valve, pressure reducer, etc if required!). Problem The following...
-
In Exercises 105108, evaluate each expression without using a calculator. log(ln e)
-
Why is it important that radioisotopes used as diagnostic tools in nuclear medicine produce gamma radiation when they decay? Why are alpha emitters not used as diagnostic tools?
-
Carbon disulfide (CS2) is a toxic, highly flammable substance. The following thermodynamic data are available for CS2(l) and CS2(g) at 298 K: (a) Draw the Lewis structure of the molecule. What do you...
-
(a) Increase [NH3], increase yield NO (b) Increase [H2O], decrease yield NO (c) Decrease [O2], decrease yield NO (d) Decrease container volume, decrease yield NO (fewer moles gas in reactants) (e)...
-
Use 10 steps of the golden-section search method to find the optimal dimensions for the cylindrical reactor vessel in Example 16.12. In that example, the dimensions of the vessel are given as the...
-
Using calculus, determine all maxima, \(m\) inima, and saddle points for the following unconstrained two-dimensional objective functions: (a) \(f\left\{x_{1} x_{2} ight\}=2 x_{1}^{3}+4 x_{1}...
-
As shown in the ASPEN PLUS simulation flowsheet in Fig. 21.15, liquid toluene is to be heated from 100 to \(350^{\circ} \mathrm{F}\) while liquid styrene is to be cooled from 300 to \(100^{\circ}...

Study smarter with the SolutionInn App


