In Prof. Patersons synthesis, the thioester (1) was deprotonated to generate an enolate (2) with defined stereochemistry,
Fantastic news! We've Found the answer you've been seeking!
Question:
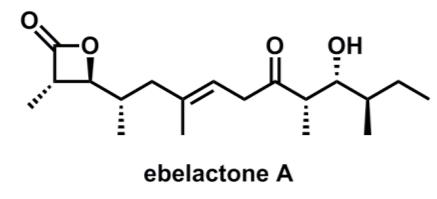
In Prof. Paterson’s synthesis, the thioester (1) was deprotonated to generate an enolate (2) with defined stereochemistry, which then participates in an aldol reaction with aldehyde (3) to give the product (4)
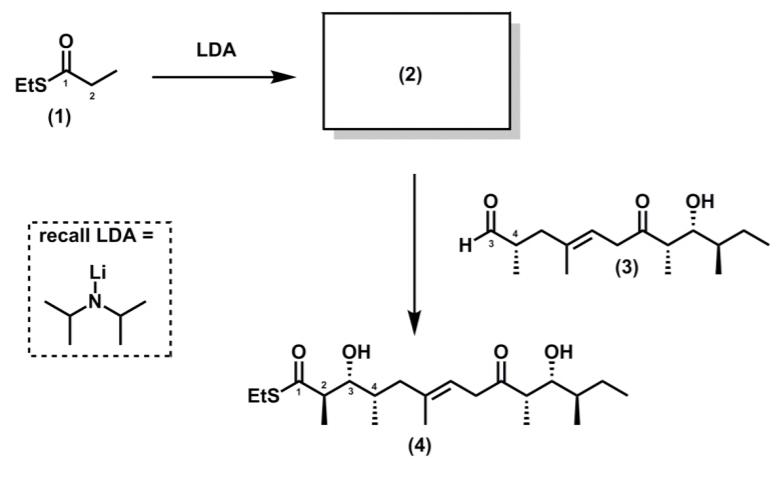
(a) Suggest a structure for enolate (2)
(b) Using cyclic deprotonation models, justify why the enolate (2) has a particular stereochemistry (E or Z).
(c) Using your knowledge of Felkin-Anh control, rationalize why compound (4) has a syn stereochemical relationship between the stereogenic centres labelled 3 and 4.
(d) Using 3D representations, invoke a Zimmerman structure to account for why compound (4) has the anti stereochemical relationship between the stereogenic centres labelled 2 and 3.
Related Book For 

Posted Date:




