Question: Label the pH scale below, indicating theacidity of a solution in the given regions. Indicate the color of phenolphthalein withinthe pH ranges shown below. Why
Label the pH scale below, indicating theacidity of a solution in the given regions.
Indicate the color of phenolphthalein withinthe pH ranges shown below.
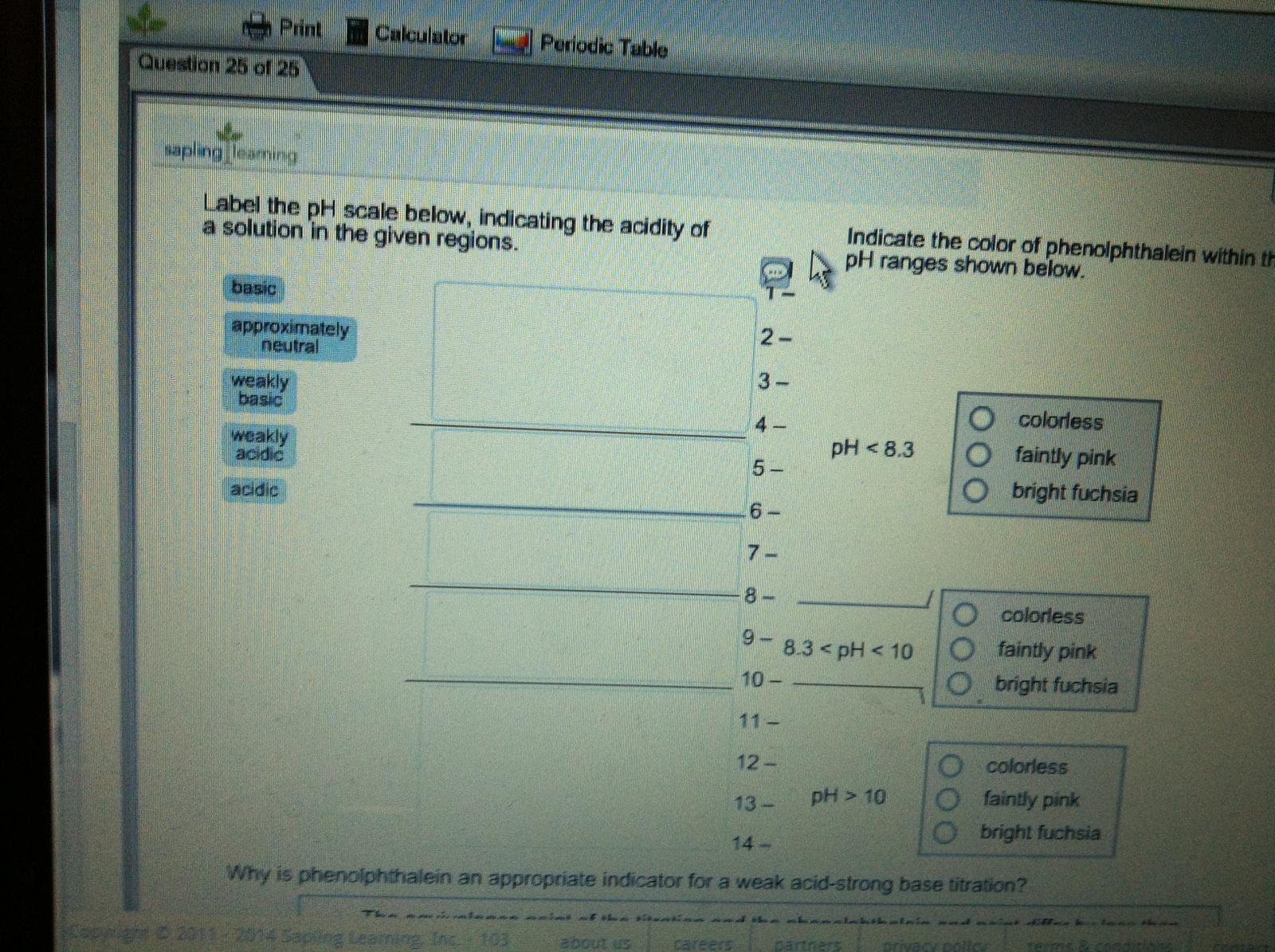
Why is phenolphthalein an appropriate indicatorfor a weak acid-strong base titration?
A. Theequivalence point of the titration and the phenolphthalein endpoint differ by less than one pH unit, so the change in colorassociated with the indicator endpoint will very closely indicatethe chemical equivalence point.
B. The equivalencepoint and the end point are both at a pH of 7, so the indicatorwill change color at the equivalence point.
C. The equivalencepoint and the end point are both at a pH of 10, so the indicatorwill change color at the equivalence point.
Question 25 of 25 basic sapling leaming Label the pH scale below, indicating the acidity of a solution in the given regions. approximately neutral weakly basic Calculator weakly acidic acidic Periodic Table Copyright 2011-2014 Sapling Learning, Inc.-103 2- !! A careers 1 5- 7- 8 1 10- 11- 12- 13- 14- Indicate the color of phenolphthalein within the pH ranges shown below. pH < 8.3 8.3 < pH < 10 pH > 10 Cartners colorless faintly pink bright fuchsia Why is phenolphthalein an appropriate indicator for a weak acid-strong base titration? and the chalkitain and The at the about us privacy pollo colorless O faintly pink Obright fuchsia colorless Ofaintly pink bright fuchsia Terms & CARE
Step by Step Solution
3.42 Rating (152 Votes )
There are 3 Steps involved in it
The detailed answer for the above question is provided below SOLUTION A scale called pH which stands ... View full answer

Get step-by-step solutions from verified subject matter experts


